Question #8e2cf
1 Answer
The answer to this one is kind of obvious, if you think about it. The volume of the balloon will decrease as more gas escapes.
This is an instance of Avogadro's Law, which states that volume and the number of moles of gas have a direct relationship when pressure and temperature are kept constant.
In other words, if temperature and pressure are constant, the more moles of a gas you have in the balloon, the larger the volumewill be. Likewise, fewer moles will imply a smaller volume.
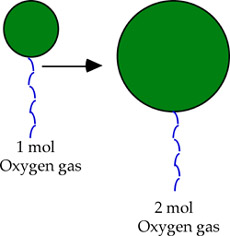
Mathematically, this is expressed as
Therefore,
Because gas is escaping the balloon,
SIDE NOTE The same principle applies when you're blowing up a balloon. Since temperature and pressure are constant, the more air you blow into the balloon, the bigger it will get.

