What is the name for #"C"_5"H"_12#? What is the condensed formula for 4-ethylnonane?
3 Answers
The name of the compound is pentane. It is an alkane.
(pent- five). There are 5 Carbon atoms, hence the name.
Explanation:
This is the condensed structure of 4-ethylnonane. As you can see, the group written in bold is the group containing the ethyl group (
Hope this helps!
The first compound is pentane.
Explanation:
Notice that the first compound does not contain any functional groups and no double or triple bonds. This implies that you're actually dealing with an alkane.
The name of the compound will thus end in -ane.
To determine its name, count the number of carbon atoms that form the parent chain. Since you have 5 carbon atoms, the compound's name will contain the prefix pent-.
So,
Now for your second compound. The name ends in -ane as well, so you're once again dealing with an alkane.
This time, however, the parent chain has more than five carbon atoms. The non- prefix corresponds to the number 9, which implies that your parent chain has nine carbon atoms.
The condensed formula for nonane looks like this
Now focus on the first part of the name. The number 4- tells you that you have a functional group added on the fourth carbon. This means that the fourth carbon will only have 1 hydrogen atom bonded.
The functional group that replaced that hydrogen is ehtyl, which looks like this
So, insert the ethyl group on that carbon to get
And there you have it - the condensed formula for 4-ethyl-nonane.
The first compound, having 5 carbons, and being an alkane, is pentane.
4-ethyl-nonane is a 9-carbon alkane with a two-carbon alkyl group (i.e. ethyl group) on carbon 4 (i.e. one away from the center carbon from either side).
Thus, you have the condensed formula:
or:
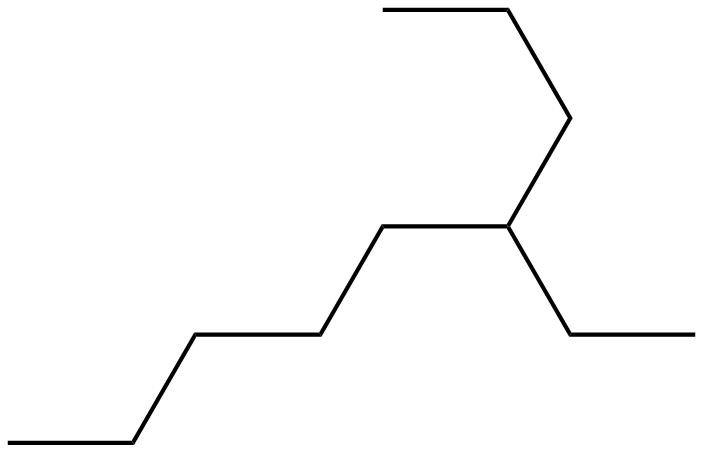
Starting from the upper middle, going down to the bottom left is the main 9-carbon chain, and on carbon 4 then is the ethyl substituent.



