Question #e121a
2 Answers
You will be left with
Explanation:
A radioactive isotope's nuclear half-life tells you how much time must pass before the half of the original sample undergoes decay.
In your case, the half-life of iodine-131 is 8 days. This means that any sample of iodine-131 that undergoes decay will be halved after 8 days, halved again after another 8 days, halved again after another 8 days, and so on.
You know that the fuel rods are stored underwater for 56 days. You can use this value to determine how many half-lives will pass.
Plug in your values to get
So, if the initial amount gets halved with every passing half-life, you can write
In your case, you have
This means that the fraction remaining after 56 days will be
1/128 remains
Explanation:
The first thing to do is count how many half lives have elapsed.
The half life (
You can see that 56 days is 7 half lives since 56/8 = 7
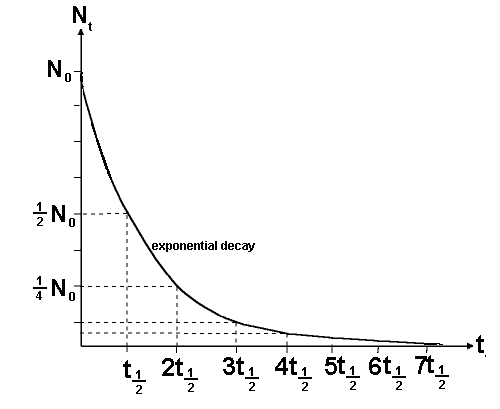
To get the fraction remaining we need to x by 1/2 seven times:
1/2 x 1/2 x 1/2 x 1/2 x 1/2 x 1/2 x 1/2 = 1/128
If you don't get nice numbers like this you can use the equation for radioactive decay:
We get the decay constant from the half life:
Taking natural logs
From which
To compare this with the other method we can check the value of 1/128 = 0.00781 so that agrees quite nicely.


