Question #6a8b4
1 Answer
Actually, chlorine has a higher electron affinity than fluorine.
Explanation:
Electron affinity is a measure of the energy released when one mole of electrons is added to one mole of atoms in the gaseous state.
In simple terms, electron affinity tells you "how bad" an atom wants to add an electron to its outermost shell.
Since electron affinity deals with energy released, the values given have a negative sign, which further emphasizes the idea that you're dealing with energy that is given off by the process.
Now, the electron affinities for fluorine and chlorine are
#EA_"F" = -"328 kJ/mol"#
#EA_"Cl" = -"349 kJ/mol"#
What this tells you is that chlorine actually has a higher electron affinity than fluorine, because it gives off more energy when it forms the chloride ion than fluorine does when it forms the fluoride ion.
Remember, the negative sign just means "given off".
Now, the question is why does chlorine have a higher electron affinity than fluorine?
Because of fluorine's smaller atomic size. Fluorine's outermost electrons are located on the second energy level, closer to the nucleus than chlorine's outermost electrons, which are located on the third energy level.
Moreover, fluorine's outermost electrons are not screened as efficiently as chlorine's outermost electrons from the nucleus' positive charge.
Despite all this, fluorine has a lower electron affinity because its outermost electrons are packed more tightly.
Fluorine is a very small atom, which means that its outermost electrons are found in a very small area.
An incoming electron will thus have to be squeezed into a very small and very negatively charged area, which implies that it will encounter significant repulsion.
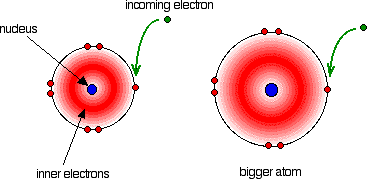
By comparison, the electron that is added to chlorine does not encounter as much repulsion because the electrons are more spread out on higher energy levels.
So, fluorine has a smaller electron affinity than chlorine because it's a smaller atom with a significant electron density concentrated in a smaller area, which means that the incoming electron must face more repulsion than it has to face in chlorine's case.

