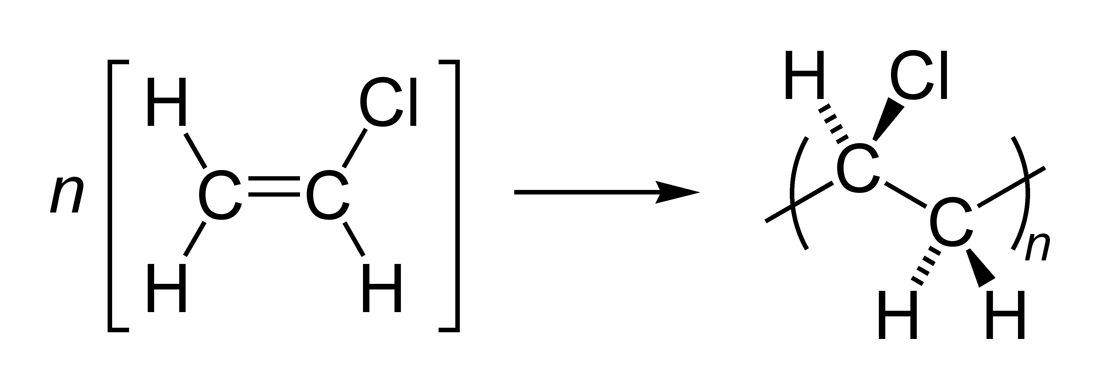Question #481a7
1 Answer
Nov 4, 2015
The correct answer is A.
Explanation:
When the polymer forms from the monomers, the reaction will occur by breaking the
Therefore, all the
Therefore, the