Question #72f8f
1 Answer
Jun 21, 2016
One sixteenth part.
Explanation:
Half life means that half of the number of atoms you start with has decayed (i.e. emitted radiation):
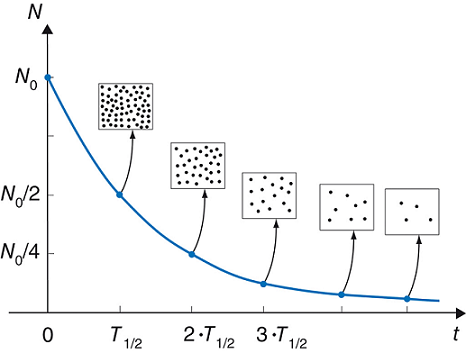
Iodine 131 has a half life of
- 1 half life = 1/2 left
- 2 half lifes = 1/4 left
- 3 half lifes = 1/8 left
- 4 half lifes = 1/16 left
For more complex calculations you can use the equation:
In which

