What are the resonance isomers for #CO_2#?
1 Answer
Feb 25, 2018
There are
Explanation:
The website actually also lists
(These are the same thing.)
The only difference between then is how the valence electrons for the two oxygens are drawn, but, in both cases, the number of valence electrons on both is the same.
The three resonance structures for
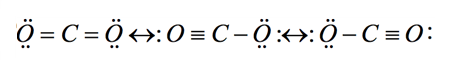
However, only one of those structures is the major resonance contributor.
For more information as to why, check out this great answer by Stefan V.!

