Question #2adff
1 Answer
They can't form hydrogen bonds/forces with water.
Explanation:
In general, for a compound to be soluble in water, it has to be able to form hydrogen bonds with water.
Also, these hydrogen bonds have to be stronger than the existing intermolecular bonds and attractions of the solute and of water.
Ionic Molecules
If we look at
Water attaches to the
Ions can only dissolve in water if the attraction between the ions and water is stronger than the attraction between the ions themselves. This is why
Polar compounds
Polar compounds can only dissolve in water if they can form hydrogen bonds with water. This is because the energy created when polar bonds are formed is less than the energy needed to break hydrogen bonds, so no bonds will form between the solute and water.
Molecules that can form hydrogen bonds with water do dissolve in water (even if they don't form hydrogen bonds between themselves). This is because when hydrogen bonds are broken in water, they can be replaced with equivalent bonds between water and the solute.
Amine dissolution in water:
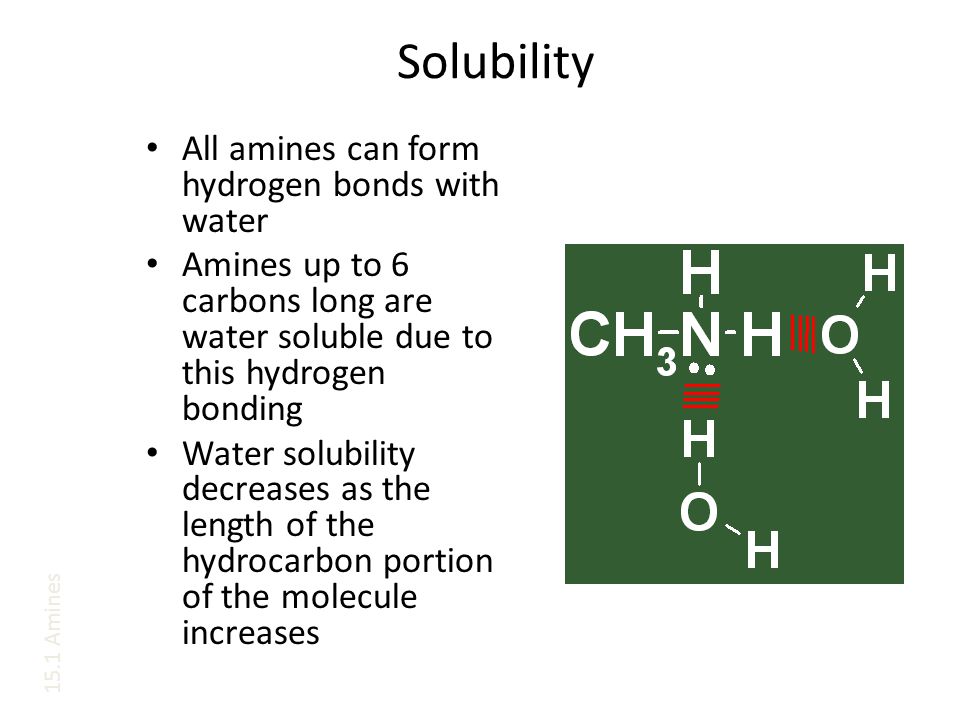
But this isn't the case with haloarenes because the energy needed to break the hydrogen bonds is higher than the energy released when new bonds form between water and the haloarene.
However, some basic haloarenes are slightly soluble in water, such as bromobenzene.

