Question #cda90
2 Answers
The best answer is d
Explanation:
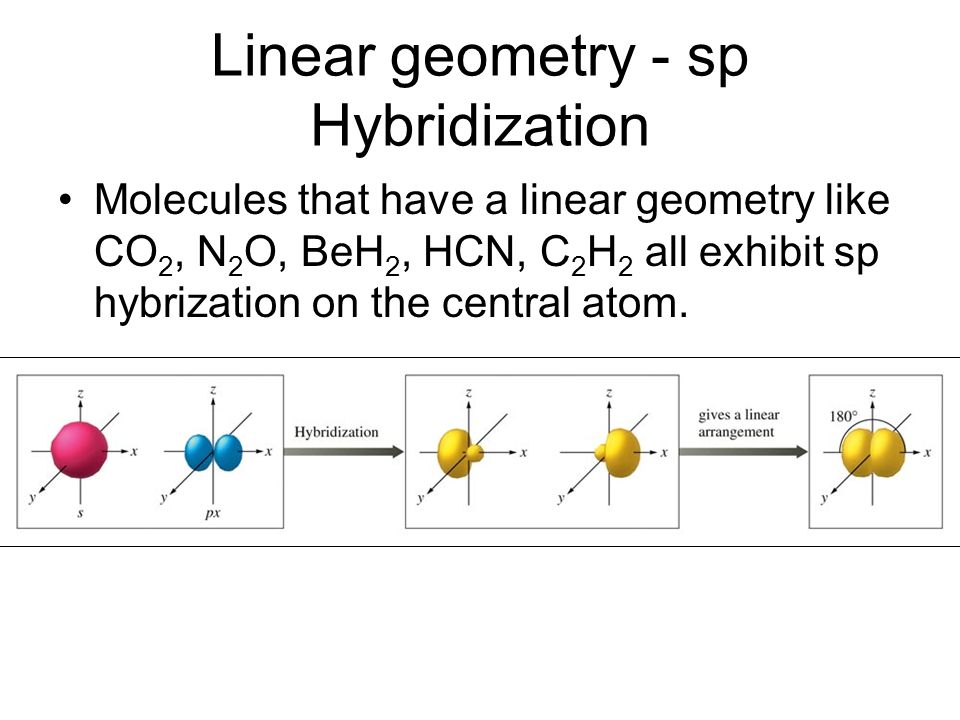
In the drawing carbon has four bonds. A single bond to the other carbon and a triple bond to the nitrogen.
A. has only two bonds so can not be the answer
B. has four bonds so could be the answer but there are two s bonds using the s orbital shown Carbon has only one s orbital
C. has only two bonds it show the s orbital being split between two p orbitals The splitting of the s orbital is correct but needs four bonds
D. has four bonds so could be correct and it shows the splitting of the s orbital between the three p orbitals (best answer)
E. has six bonds not four. It shows the splitting of the s orbital between 4 p orbitals Carbon has only 3 p orbitals.
The electron domain geometry is linear; the
involves c)
Explanation:
Per VSEPR theory, a carbon atom that has two electron domains should have a linear electron geometry.
 Linear
Linear
(From StudyBlue)
The molecule will be linear with a bond angle of 180°.
The nitrogen atom also has two electron domains: the lone pair and the triple bond to the carbon.
Linear electron geometry corresponds to
 images.slideplayer.com
images.slideplayer.com
Since both

