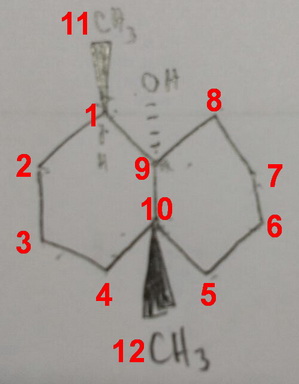
The name of your compound is 1,10-dimethylbicyclo[4.4.0]decan-9-ol.
For easy reference, I have inserted the numbering for you.
The chiral centres appear to be carbons 1, 9 and 10 (all the others have at least two #"H"# atoms, so they can't be chiral).
Configuration at #"C1"#:
#"C1"# is attached to #"C2, C9, C11"#, and #"H"#.
#"H"# is obviously Priority 4, but the carbon atoms are all tied. We must go one atom further out from each atom to break the tie.
#"C2"# is attached to #"C3, H"#, and #"H (C,H,H)"#.
#"C9"# is attached to #"O, C8"#, and #"C10 (O,C,C)"#.
#"C11"# is attached to #"H, H"#, and #"H (H,H,H")#.
We compare these atoms at the first point of difference.
Since #"O > C >H"#, #"C9"# is Priority 1, #"C2"# is Priority 2, and #"C11"# is Priority 3.
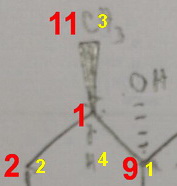
#"H"# is in the rear, and the #1 → 2 → 3# direction is clockwise, so #"C1"# has the (#R#) configuration.
Configuration at #"C9"#:
#"C9"# is attached to #"O, C1, C8"#, and #"C10"#.
#"O"# is obviously Priority 1, but the carbon atoms are all tied.
#"C1"# is attached to #"C2, C11"#, and #"H (C,C,H)"#.
#"C8"# is attached to #"C7, H"#, and #"H (C,H,H)"#.
#"C10"# is attached to #"C4, C5"#, and #"C12 (C,C,C)"#.
#"C10 (C,C,C) > C1 (C,C,H) > C7 (C,H,H)"#, so #"C10"# is Priority 2, #"C1"# is Priority 3, and #"C8"# is Priority 4.
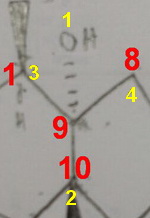
If you view along the #"C9-C8"# bond with the #"C9-C10"# bond in the plane of the paper, #"C1"# comes in front of the paper.
The #1 → 2 → 3# direction is counterclockwise, so the configuration at #"C9"# is (#S#).
Configuration at #"C10"#:
#"C10"# is attached to #"C4, C5, C9"#, and #"C12"#.
#"C4"# is attached to #"C3, H"#, and #"H (C,H,H)"#.
#"C5"# is attached to #"C6, H"#, and #"H (C,H,H)"#.
#"C9"# is attached to #"O, C3"#, and #"C8 (O,C,C)"#.
#"C12"# is attached to #"H, H"#, and #"H (H,H,H)"#.
#"C9 (O,C,C) > C12 (H,H,H)"#, so #"C9"# is Priority 1 and #"C12"# is priority 4.
To break the tie between #"C4"# and #"C5"# for Priorities 2 and 3, we move out another three carbons.
That brings #"C4"# to #"C1 (C,C,H)"# and #"C5 to C8 (C,H,H)"#.
Thus, #"C4"# is Priority 2, and #"C5"# is Priority 3.

The #1 → 2 → 3# direction is clockwise, so we would assign the configuration (#R#).
However, the lowest priority group is in front, so we reverse the assignment to (#S#).
The compound is [#1R,9S,10S#]-1,10-dimethylbicyclo[4.4.0]decan-9-ol.





