Finding R and S for Chiral Centers
Key Questions
-
It is a stereochemical label to indicate the relative spatial orientation of each atom in a molecule with a non-superimposable mirror image.
R indicates that a clockwise circular arrow that goes from higher priority to lower priority crosses over the lowest priority substituent and that lowest-priority substituent is in the back.
The R and S stereoisomers are non-superimposable mirror images, which means if you reflect them on a mirror plane, they do not become the exact same molecule when you overlay them.
When you label a molecule as R or S, you consider the priorities of each substituent on the chiral carbon (connected to four different functional groups).
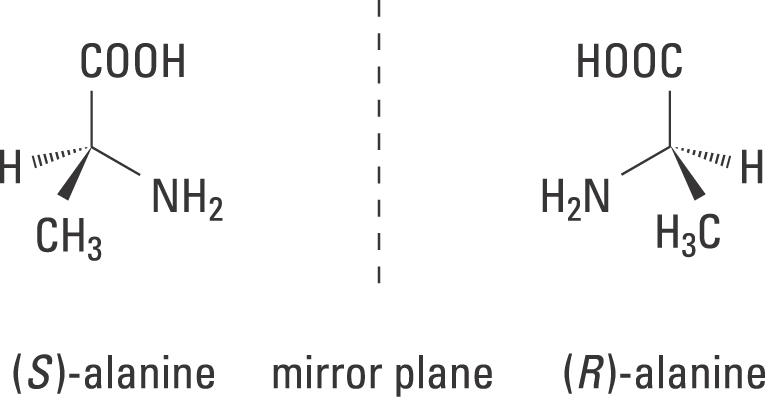
Let's take this chiral amino acid for example:
Some general ways you could determine the priorities are:
- HIgher atomic number of the directly-attached atom gives higher priority
- Atomic number of the atom attached to the one is considered in step 1 if two substituents have the same first atom
- Higher number of same-atom branches determines greater priority if the overall substituents are too similar (e.g. isopropyl has higher priority than ethyl)
With (R)-alanine:
#"NH"_2# has priority 1 due to highest atomic number for#"N"# .#"COOH"# has priority 2 due to the higher atomic number of#"O"# vs.#"H"# in#"CH"_3# #"CH"_3# has priority 3 as a result.#"H"# has priority 4.
Now, if you draw a circular arrow starting at
#"NH"_2# , going to#"COOH"# , crossing over#"H"# since it is in the back, and to#"CH"_3# , then you would have gone clockwise.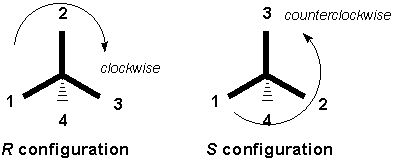
Since the lowest priority atom is in the back, the clockwise arrow corresponds to the R configuration.
If you had started from the same R configuration but oriented
#"H"# in the front and#"CH"_3# in the back, it would have been S configuration. Let's call this S configuration A, where you just nudge two substituents to flip them from front/back to back/front.If you reflect the same R configuration over a mirror plane, keeping the orientations of
#"H"# in the back and#"CH"_3# in the front after the flip, the configuration is also S. Let's call this S configuration B, where you've actually done a reflection.If you start from S configuration B, and flipped it over a vertical axis (literally rotating
#180^o# in space), you would get S configuration A. -
R and S are used to describe the configuration of a chirality center. Chirality center meaning that there are 4 different groups attached to one carbon. To determine whether the chirality center is R or S you have to first prioritize all four groups connected to the chirality center. Then, rotate the molecule so that the fourth priority group is on a dash (pointing away from you). Finally, determine whether the sequence 1-2-3 is (R) clockwise or (S) counterclockwise. Hope this helps.