Question #085e9
1 Answer
Sep 10, 2017
It is because ionic size increases from top to bottom in Group 1.
Explanation:
All alkali metals react with hydrogen to form metallic hydrides:
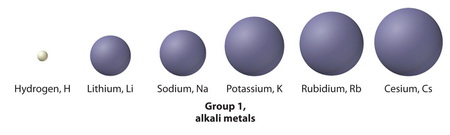
The closer you get to the bottom of Group 1, the larger the atom becomes.
Thus, a
This makes

