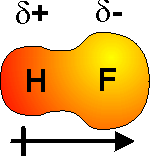Question #b1d48
1 Answer
Here's how you can do that.
Explanation:
Hydrogen fluoride molecules,
- the hydrogen atom develops a partial positive charge as a result of the difference in electronegativity between hydrogen and fluorine
- the fluorine atom has
#3# lone pairs of electrons
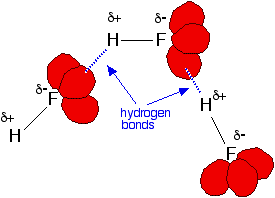
As you might suspect, hydrogen bonds are formed between the partial positive hydrogen atom and the lone pairs present on the fluorine atom of a neighboring hydrogen fluoride molecule.
More specifically, each partial positive hydrogen can form a hydrogen bond with one lone pair of electrons present on the fluorine atom of a neighboring hydrogen fluoride molecule.
Now, to draw the dipole of a hydrogen fluoride molecule, draw an arrow with a plus sign starting at the hydrogen atom and pointing towards the fluorine atom.
The partial positive charge,
Here's how molecules of hydrogen fluoride would look like with hydrogen bonds connecting them
Notice that each molecule of hydrogen fluoride can form just one hydrogen bond at given time due to the fact that each molecule has only one partial positive hydrogen atom.
So, to draw a hydrogen bond between two hydrogen fluoride molecules, simply draw a dotted line that goes from the partial positive hydrogen atom of one molecule to one of the