What is the effective atomic number of dichlorobis(ethylenediamine)cobalt(III)?
1 Answer
#36# .
The effective atomic number is an old term meaning the sum of ALL the electrons for the central atom and the donated valence electrons from the surrounding ligands, using the appropriate electron-counting scheme.
We will use something I call the "oxidation state" counting scheme, wherein each single-oxidation-state atom is taken as its common oxidation state, and the sum of the charges adds up to the charge of the complex.
#["Co"("en")_2"Cl"_2]^(+)#
has oxidation states as
#[stackrel(+3)"Co"" "stackrel(" "2(0))(("en")_2)stackrel(" "2(-1))("Cl"_2)]^(+)# ,since we take
#"en"# , ethylenediamine, to be a neutral#sigma# donor, and each#"Cl"# ligand to be the#"Cl"^(-)# #pi# donor.
- Thus,
#2(-1) + [?] = +1# , and#[?] = +3# for cobalt cation, which means a total of#27 - 3 = bb24# electrons for#"Co"^(3+)# . #"Cl"^(-)# in compounds donates two valence electrons as#bb( : )"Cl"^(-)# .#"en"# ,
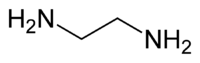
a bidentate ligand, donates two electrons from each (of two) nitrogen heads as
#"H"_2stackrel(|)"N":# , for a total of four valence electrons per ligand.
Indeed,
That gives

