What is the osmotic pressure for #"1.95 g"# of sucrose dissolved in #"150 mL"# solution at #25^@ "C"#? #"FW"# #=# #"342.2965 g/mol"#
1 Answer
#Pi = "0.9292 atm"# .
You can read more about osmotic pressure (and other colligative properties) here.
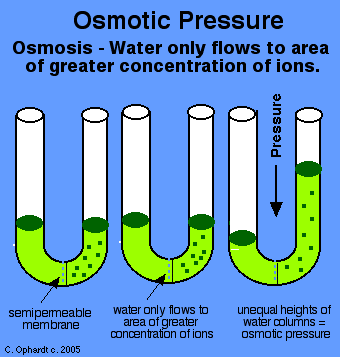
The osmotic pressure
#Pi = icRT# ,where:
#i# is the van't Hoff factor, as seen in freezing point depression and boiling point elevation. It can be approximated by the number of particles per formula unit in solution.#c# is the concentration in whatever units are appropriate. In this case, if#Pi# is in#"atm"# , then#c# is in#"mol/L"# when...- ...the universal gas constant
#R# is#"0.082057 L"cdot"atm/mol"cdot"K"# .#T# is the temperature in#"K"# .
We are indeed given the correct info to calculate the molarity of the sucrose solution.
#1.95 cancel"g suc" xx "1 mol suc"/(342.2965 cancel"g suc")#
#=# #"0.005697 mols suc"#
So, the molarity is:
#c = "0.005697 mols suc"/(150 xx 10^(-3) "L soln")#
#=# #"0.03798 M"#
Therefore, the osmotic pressure at
#color(blue)(Pi) = (1)(0.03798 cancel"mol/L")(0.082057 cancel"L"cdot"atm/"cancel"mol"cdotcancel"K")(298.15 cancel"K")#
#=# #color(blue)("0.9292 atm")# ,
since nonelectrolytes have

