How does an antibonding orbital differ from a bonding orbital? How do they relate to bond order?
1 Answer
A molecular orbital (or further orbitals formed by molecular orbitals) can be bonding or antibonding. The electron is identified as being in either a bonding or antibonding molecular orbital, and is then classified as bonding or antibonding.
An antibonding orbital has one more node than its corresponding bonding orbital.
Bonding molecular orbitals (MOs) form due to in-phase atomic orbital overlap. Antibonding MOs are due to out-of-phase overlap.
The most fundamental kinds of overlaps are sigma/
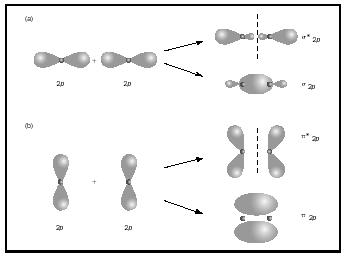
- Figure
#(a)# shows the#sigma_(2p)^"*"# antibonding MO and the#sigma_(2p)# bonding MO to the right. - Figure
#(b)# show the#pi_(2p)^"*"# antibonding MO and#pi_(2p)# bonding MO to the right.
Most typically, an orbital with a
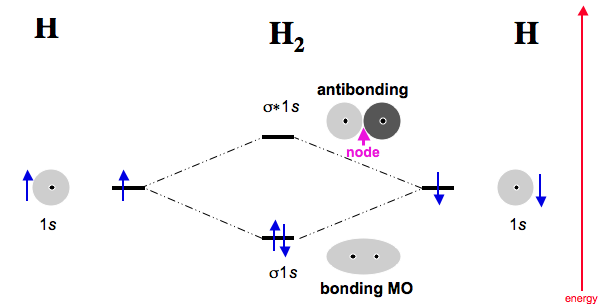
In this case, there are two electrons in the
#"BO" = 1/2("Bonding e"^(-) - "Antibonding e"^(-))#
#= 1/2(2 - 0) = 1# ,
as it should be for a single bond in

