This is zinc peroxide (not "zinc(IV)" oxide!), i.e. it contains #2 xx "O"# in the #color(red)(-1)# oxidation state.
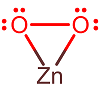
The way you would know that is by already knowing that zinc has a maximum oxidation state of #+2#. Its electron configuration is #[Ar] 3d^10 4s^2#, so losing more than two electrons is nearly impossible (it would require freeing up a quantum state in the #3d# outer-core orbitals).
Hence, the zinc atom has an oxidation state of #color(blue)(+2)# (not #+4#, and not #+1#).
You may accidentally say that this is simply #2 xx "O"^(2-)# for the anion(s). You may also miss that for the anion without prior knowledge or without looking it up, it is a non-innocent ligand that could either be #"O"_2^(-)# (the superoxide anion) or #"O"_2^(2-)# (the peroxide anion).
Either misconception could lead you to accidentally say that zinc has an oxidation state of #+4# or #+1#. Neither is correct!


