How can an electron have a node?
1 Answer
Aug 22, 2017
Well, an electron has wave-like properties.......
Explanation:
And a node is a point on a wave-function of MINIMUM displacement.
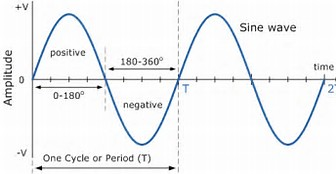
The nodes on the given wave-function (a sine wave),

