A sample of gas at a pressure of 121.59 kPa, a volume of 31 L, and a temperature of 360 K contains how many moles of gas?
1 Answer
Sep 17, 2016
The sample of gas contains 1.3 moles of gas.
Explanation:
Use the ideal gas law as shown below.
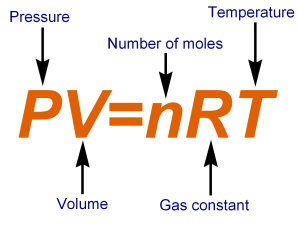
Rearrange the equation to isolate

