Describe all 4 quantum numbers for last electron of 3d^5?
2 Answers
see below
Explanation:
it is Mn (manganese)
N= 3 third energetic level
l= 3 shape of orbitals of type d : (quatrefoil)
5 = the 5 orbitals d are all full means with only one electron in every orbitals (M,magnetic orbitatals = -2,-1, 0 +1 +2 what is the last, you don't know because are isoenergetic)
s =intrinsic magnetic moment
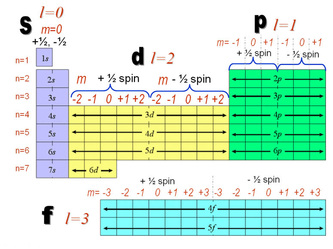
The four quantum numbers are 3, 2, +2, +½.
Explanation:
The four quantum numbers are
#n color(white)(ll)=# principal quantum number; determines energy and size of orbital#l color(white)(m)= # secondary quantum number; determines shape of orbital#m_text(l) =# magnetic quantum number; determines orientation of orbital in magnetic field#m_text(s) =# spin quantum number; determines direction of electron spin
The rules for quantum numbers are:
#n color(white)(ll)=# integers from 1 to ∞.#lcolor(white)(m)= # integers from 0 to#n - 1# #m_text(l) =# integers from#-l# to#+l# #m_text(s) =# +½ or -½
Here is a table of quantum numbers for the first five
#ulbb(ncolor(white)(m)lcolor(white)(ml)m_text(l)color(white)(ml)m_text(s))#
#3color(white)(m)2color(white)(mll)"-2"color(white)(m)"+½#
#3color(white)(m)2color(white)(mll)"-1"color(white)(m)"+½#
#3color(white)(m)2color(white)(mm)"0"color(white)(m)"+½#
#3color(white)(m)2color(white)(m)"+1"color(white)(m)"+½#
#color(red)(3color(white)(m)2color(white)(m)"+2"color(white)(m)"+½)#
Here's how the quantum numbers fit in the Periodic Table