How are density mass and volume related?
1 Answer
They are related by the the density triangle.
Explanation:
They are related by the the density triangle.
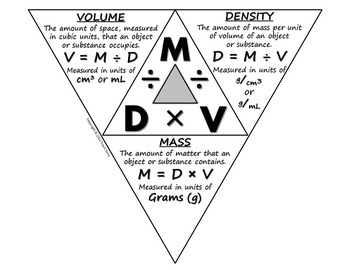
d =
m = d×V
V =
DENSITY
Density is defined as mass per unit volume.
d =
Example:
A brick of salt measuring 10.0 cm x 10.0 cm x 2.00 cm has a mass of 433 g. What is its density?
Step 1: Calculate the volume
V = lwh = 10.0 cm × 10.0 cm × 2.00 cm = 200 cm³
Step 2: Calculate the density
d =
MASS
d =
We can rearrange this to get the expression for the mass.
m = d×V
Example:
If 500 mL of a liquid has a density of 1.11 g/mL, what is its mass?
m = d×V = 500 mL ×
VOLUME
d =
We can rearrange this to get the expression for the volume.
V =
Example:
What is the volume of a bar of gold that has a mass of 14.83 kg. The density of gold is 19.32 g/cm³.
Step 1: Convert kilograms to grams.
14.83 kg ×
Step 2: Calculate the volume.
V =

