How can I represent an endothermic reaction in a potential energy diagram?
1 Answer
Dec 20, 2015
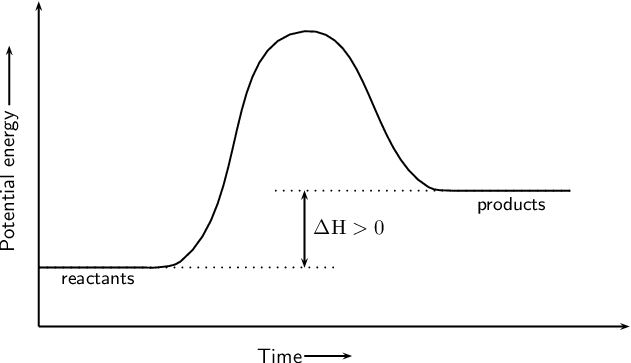
An endothermic reaction must have a positive change in enthalpy. That is,
To represent the positive change in enthalpy, the potential energy of the reactants must be LOWER than that of the products. Notice how the end of the "hump" is higher than the beginning, showing how the system gained energy during the course of the reaction.

