How can water change state?
1 Answer
Water can change state by gaining or losing energy.
Explanation:
If gaseous water loses energy (heat), the kinetic energy or speed of the molecules slow down. When the temperature of the water molecules reaches 100º C, temporary bonds caused by inter-molecular attraction can form. This attraction between water molecules join the molecules loosely together and they become a liquid.
The reverse is also true. When liquid water reaches 100º C, the temporary bonds between the molecules break apart from each other and become gaseous molecules.
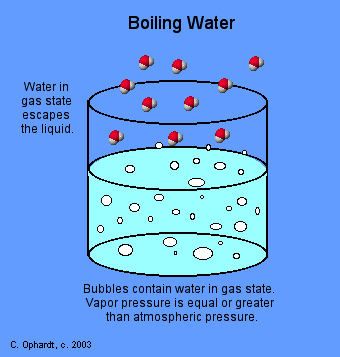
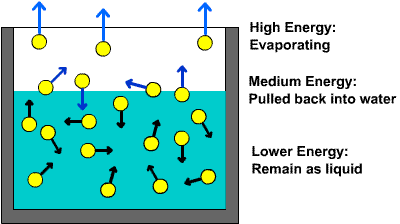
Water does not have to be at 100º C to become a gas, however. Individual molecules can break the bonds that make them a liquid and enter the air at any temperature above 0º C. This is called evaporation. More Information
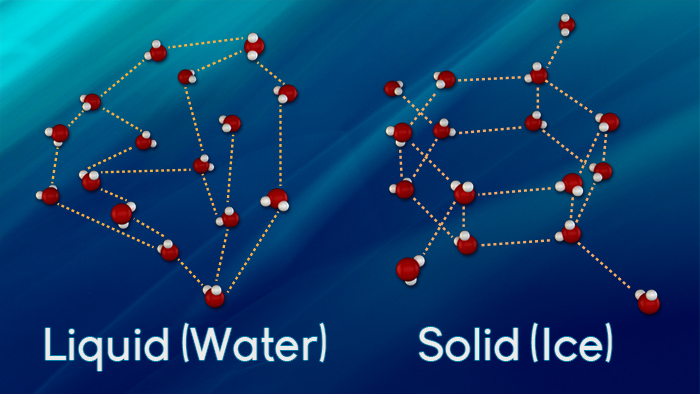
Liquid water turns into solid water at 0º C. As the molecules lose more energy, more of the temporary bonds between the molecules form. When a group of six water molecules are joined by hydrogen bonds ( the attraction between molecules) they form an ice crystal. Below 0º C, of the water molecules have been joined together to form a solid.
The reverse is also true. At 0º C, the bonds are broken. As more energy is added, the bonds of intermolecular attraction are broken faster than they are formed and the ice crystals break apart. The ice turns into liquid water.