How can we know that which element is more electronegative than other ? I've never come across any such formula or trick for this,except learning it by practice. Please tell if you know any.
Eg: in heterolytic fission of CO, we know that oxygen will take both the electrons but what if there was some other element whose relative electronegativity I dont know, then how can I solve it.
Eg: in heterolytic fission of CO, we know that oxygen will take both the electrons but what if there was some other element whose relative electronegativity I dont know, then how can I solve it.
2 Answers
Electronegativity INCREASES across a Period, from left to right as we face the Table...........
Explanation:
And excluding the
Why should this be so? Electronegativity tends to be a function of NUCLEAR charge (and thus dependent on
Down a Group, the
And so let's have a gander at the Pauling Scale, and examine the electronegativities:
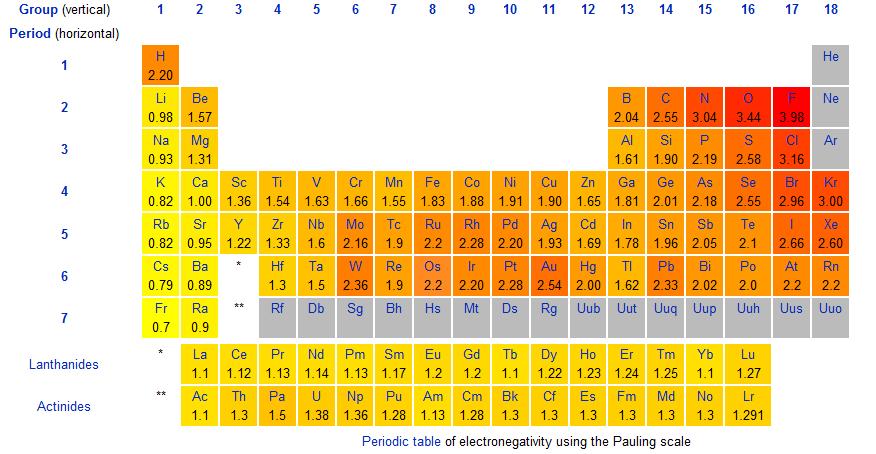
Is this representation roughly consistent with what I have argued? The most electronegative element should thus be fluorine, and indeed it is. On the other hand, the LEAST electronegative elements should be the alkali, and alkaline-earth metals. Is this true?
This is one of the most important trends a chemistry undergraduate can learn. If you are unsatisfied with this answer, or seek clarification, voice your objection, and someone will address the issue.
And thus in your example, for
Actually there are 4 or 5 formulae for calculating electronegativity according to different models.....
Explanation:
Pauling, Allen, Mullikan, Sanderson and Allred-Rochow all came up with models from which electronegativity values can be derived using formulae. None of them are especially "simple" calculations, and you will need a decent chemical data book to hand to find the various values to plug in. However, it's certainly do-able.
Wikipedia actually lists the various methods and explains how to do them here en.wikipedia.org/wiki/Electronegativity#Methods_of_calculation


