How do enzymes alter the rate of chemical reactions?
1 Answer
Enzymes speed up the rate of chemical reaction.
Explanation:
Enzymes are biological molecules made of proteins that help speed up the rate of a chemical reaction: either it serves as a catalyst, or it binds with other molecules to produce a new molecule that helps hasten a reaction. However, enzymes are highly selective - meaning, it only helps speed up a specific chemical reaction. In other words, an enzyme does not alter ALL chemical reactions, just a few select ones.
The simplest example would your saliva. The saliva contains many enzymes.
One of the enzymes is called amylase, which breaks down the food we eat into simpler units (i.e. starch into simple sugars) , which in turn is further broken down by our digestive system.
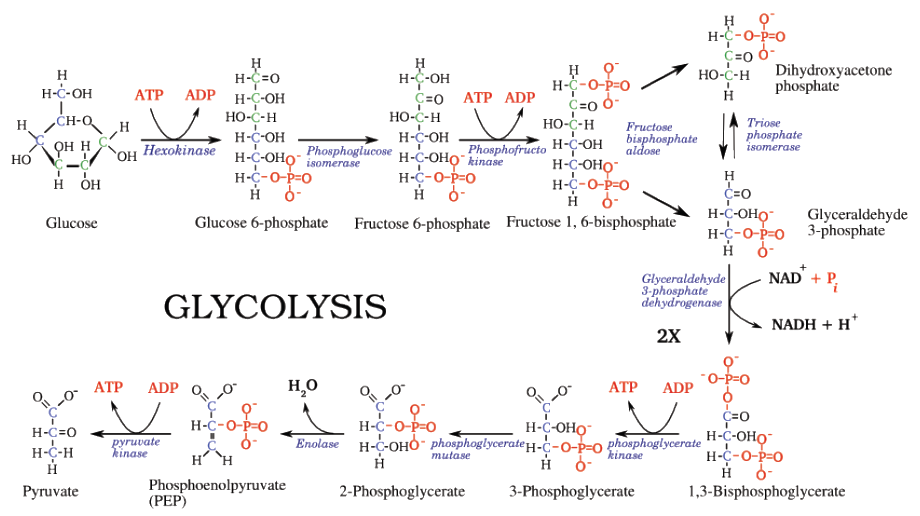
But the example that most biochemistry professors teach their students when talking about enzymes is Glycolysis (or the process of changing the digested sugar into energy or ATP). This process is a sequence of ten enzyme-catalyzed reactions (as shown in the illustration below, the enzymes are labelled in
Notice that no enzyme was repeated during the whole process since the enzyme used in each reaction is specific or is especially made for that reaction.