How do you write the orbital diagram for phosphate?
1 Answer
Aug 11, 2017
I interpreted this as the molecular orbital diagram... and it became rather complicated (I honestly don't think you want to know the minor details!).
Here's the scratchwork when I was getting the reducible representations
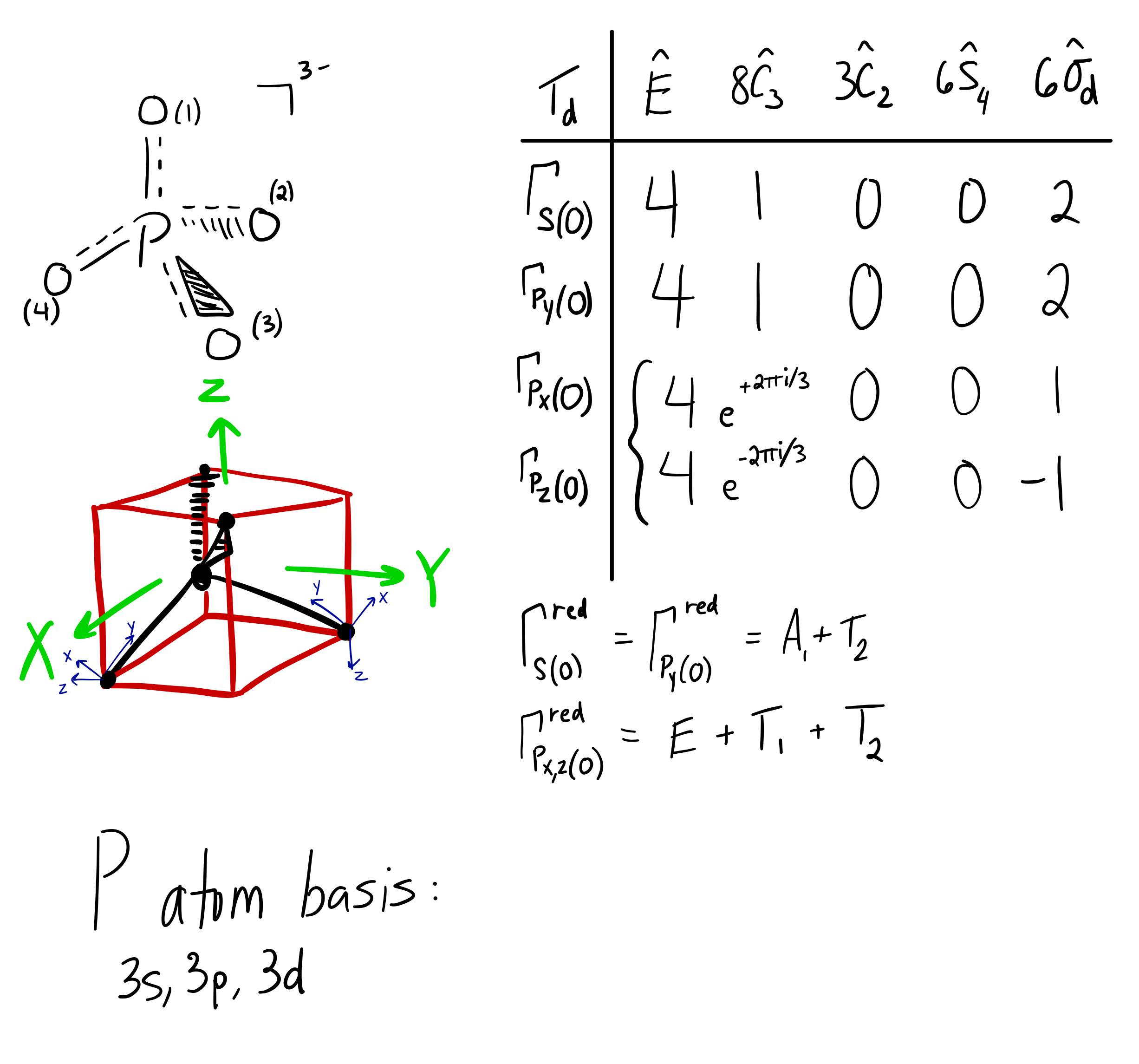
The whole point of that was to see how the oxygen orbital energies split up (and which were two-fold or three-fold degenerate). It was also to figure out which orbitals on phosphorus interact with which orbitals on the oxygen atoms.
The resultant MO diagram was:
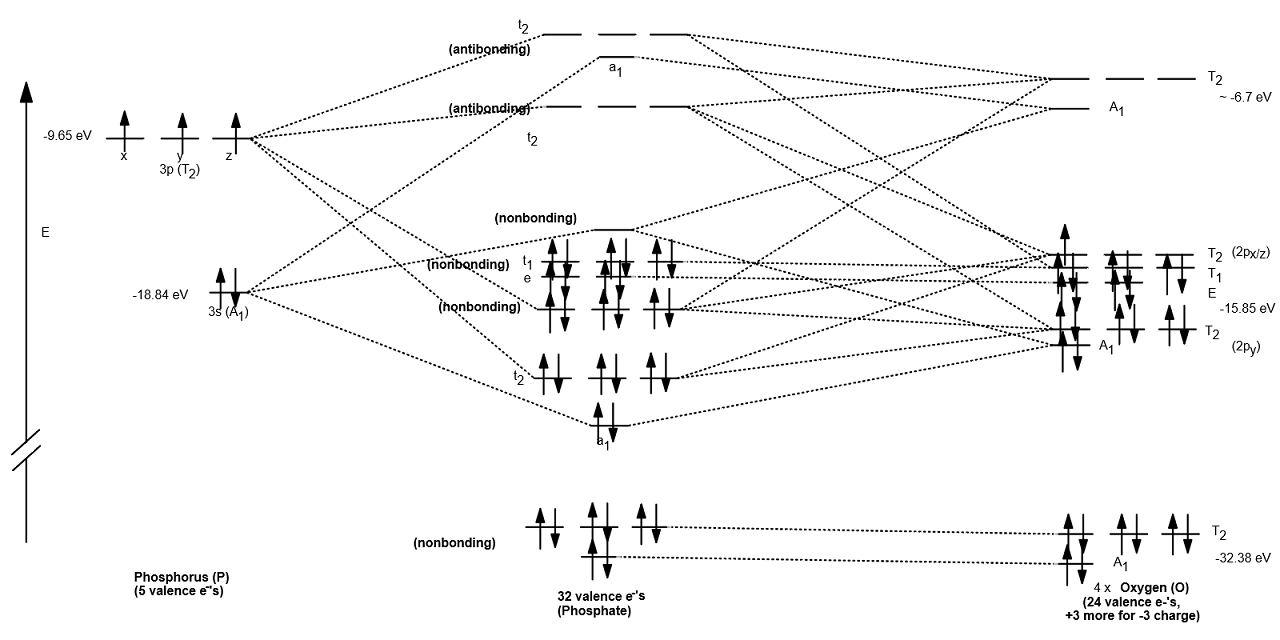
Takeaways:
- This is only qualitative, so take it with a grain of salt.
- Phosphate has all electrons paired.
- The nonbonding orbitals are nonbonding either due to mismatched symmetries, overly different inner-atom/outer-atom orbital energies, or interactions between both high-lying and low-lying orbitals.
- There are
#22# nonbonding electrons, which accounts for all#11# lone pairs of electrons that belong to the oxygen atoms. The remaining#2# nonbonding electrons account for the#bb(pi)# bond that is delocalized. - The
#sigma# bond order that this MO diagram seems to account for is#1# for all four#bb(sigma)# bonds, as there are four clearly-bonding MOs (#a_1 + t_2# ), one for each bond.

