How do you name coordination compounds? (Undergraduate level)
(Just the most common names/titles or prefixes/suffixes, please)
(Just the most common names/titles or prefixes/suffixes, please)
1 Answer
For relatively simple compounds, it is basically alphabetical ordering of ligands, with prefixes just like in general chemistry.
The main differences are that things connected to the central atom can be more complicated than simple atoms, and that there are new prefixes for complicated ligands to make names look nicer.
DISCLAIMER: REFERENCE ANSWER!
See the bottom of the answer (Appendix I, II) for prefixes and lists of ligands to know or be aware of.
SIMPLIFIED NOMENCLATURE RULES
- List ligands alphabetically.
- List the metal name last, along with its oxidation state.
- As usual, put the cation name first and put a space after it.
- If the metal is within an anionic complex, then use the name of the metal that matches the periodic table symbol and end with "-ate".
- Use prefixes from table
#"I"# (Appendix I) to indicate multiple identical simple ligands.
ISOMERIZATION RULES
There are also isomers, like cis/trans, delta/lambda, and facial/meridional. Maybe you don't need to know it, but this answer goes into more detail on that if you need it.
BINDING RULES
There are cases where one atom connect two others (bridging,
SIMPLE (UNDERGRADUATE) EXAMPLES
#["Ag"("NH"_3)_2]^(+)#
- diamminesilver(I)
Note that ammine has two#m# 's, but ethylenediamine has one#m# . The "di" indicates two ammine ligands.
#["Ti"("CO")_6]^(2-)#
- hexacarbonyltitanate(II)
Note that the ending is titanate, not titanium, because the complex has a#2^-# charge. "Hexa" indicates six identical ligands.
#"K"_4["Fe"("CN")_6]#
- potassium hexacyanoferrate(II) [not ironate]
Note the similarity to general chemistry ionic compound naming. The cation name is placed first, with a space. Also, the name of iron as seen in the periodic table symbol is used (#"Fe"^(2+//3+) harr "ferro/i"# ), not ironate.
APPENDIX I --- PREFIXES
TABLE I
#ul("Prefix"" "" ""Number of Identical Ligands")#
#"N/A"" "" "" "1#
#"di-"" "" "" "color(white)(i.)2#
#"tri-"" "" "" "color(white)(ii)3#
#"tetra-"" "color(white)(i.....)4#
#"penta-"" "color(white)(i....)5#
#"hexa-"" "color(white)(i.....)6#
#ul(vdots" "" "" "color(white)(i.)vdots" "" "" "" "" "" "" "" "" "" ")#
You probably won't see any further than hexa, just purely due to the ability of the metals to bind to ligands.
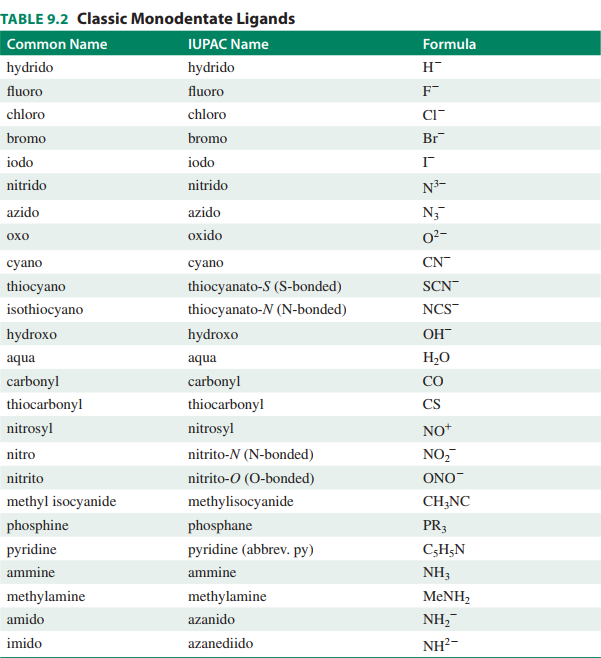
APPENDIX II --- LIGAND LIST
Here is a list of common monodentate ligands. You should ask your professor which ligands you do NOT need to know.
I would stick to the common name when naming.