How does ozone protect us?
1 Answer
Protects us from harmful UVB and UVC radiation.
Explanation:
In the Stratosphere the ozone layer absorbs harmful UVB and UVC radiation, however allowing UVA (and small amount of UVB), for photosynthesis and vitamin D, which are both essential for life.
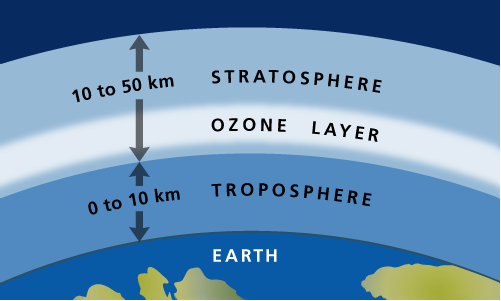
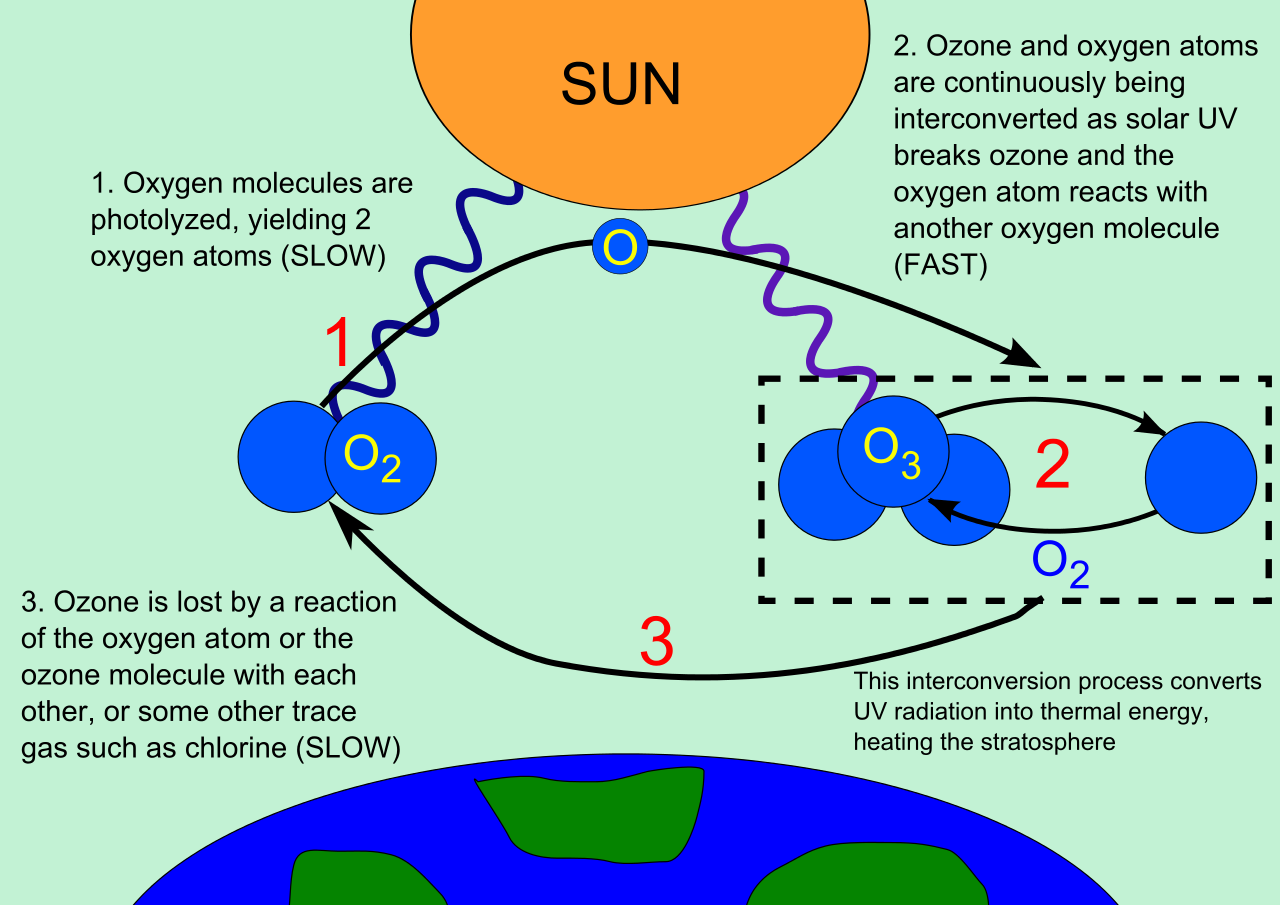
Every time an ozone reacts with UV light, it absorbs it. This is due to the process known as the 'Ozone–oxygen cycle':
-
An oxygen molecule undergoes photodissociation by high frequency UV. This creates to oxygen free radicals.
-
#O_2(g) rarr 2O·(g)# -
#O_2(g) + O·(g) rarr O_3(g)# -
The ozone molecule absorbs UVB and UVC radiation, dissociates into oxygen and oxygen free radicals.
-
#O_3(g) rarr O·(g) + O_2(g)# -
Any remaining oxygen free radicals are removed by reacting with remaining ozone. This creates oxygen gas, and thus the cycle can start over.
-
#O_3(g) + O·(g) rarr 2O_2(g)#
If there was no ozone, an increased penetration of UVC and UVB would increase skin cancer and damage plants and animals. High UV penetration can become the catalyst for the formation of photochemical smog, which occurs in the Troposphere.


