How many chiral carbon atoms are there in the open-chain form of glucose?
1 Answer
Aug 11, 2015
There are four chiral carbon atoms in the open-chain form of glucose.
Explanation:
The structure of glucose with its numbering is
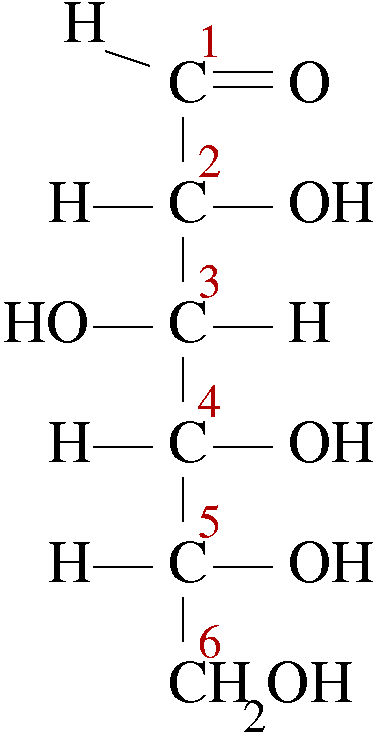
There are no internal mirror planes, so every carbon atom is different.
The four chiral centres (with their attached groups) are:
#"C2 (OH,C1, C3, H)"# #"C3 (OH, C2, C4, H)"# #"C4 (OH, C3, C5, H)"# #"C5 (OH, C4, C6, H)"#

