How many sp2 and sp3 hybridized carbons are in naproxen?
1 Answer
Apr 24, 2016
There are eleven
Explanation:
The structure of Naproxen is
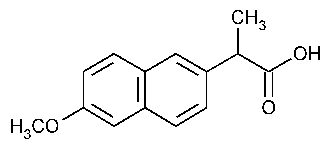
Every double-bonded carbon atom is
That includes the ten carbon atoms in the naphthalene rings and the carbonyl carbon in the carboxylic acid (11 carbon atoms).
The two methyl groups and the carbon atom attached to the ring are

