How would you describe the distribution of charge in an atom?
1 Answer
Jun 28, 2018
See explanation.
Explanation:
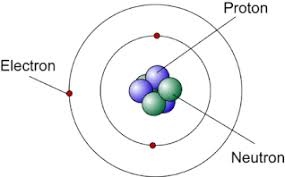
There are 2 places where charge is located in the atom:
-
the nucleus contains neutrons (zero charge) and protons (positive charge)
-
Around the nucleus there are electrons located on electron shields and the charge of electrons is equal to the charge of protons in the nucleus, but have negative sign.
To sum it all up we can say that the charge of protons in the nucleus is reduced by the charge of electrons on the orbits to make the atom electrically neutral.
However if an atom gives an electron away or gets one from another atom, then the positive and negative charges are not equal and the atom becomes an ion.
