How would you draw and label energy diagrams that depict the following reactions, and determine all remaining values? Place the reactants at energy level zero
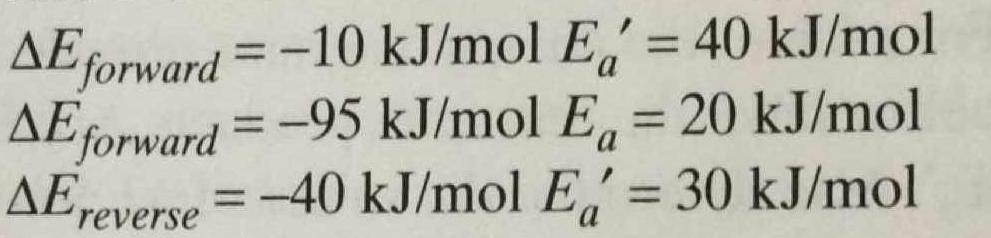
1 Answer
We would split up the given values in terms of thermodynamics and kinetics for the reactions.
Thermodynamics
#DeltaE'_(fwd) = -"10 kJ/mol"#
#DeltaE'_(rev) = -"40 kJ/mol"#
#DeltaE_(fwd) = -"95 kJ/mol"#
Kinetics
#E_a' (fwd) = "40 kJ/mol"#
#E_a' (rev) = "30 kJ/mol"#
#E_a (fwd) = "20 kJ/mol"#
The thermodynamic values correspond to the reactants vs. products. The kinetic values correspond to the reactants vs. transition state.
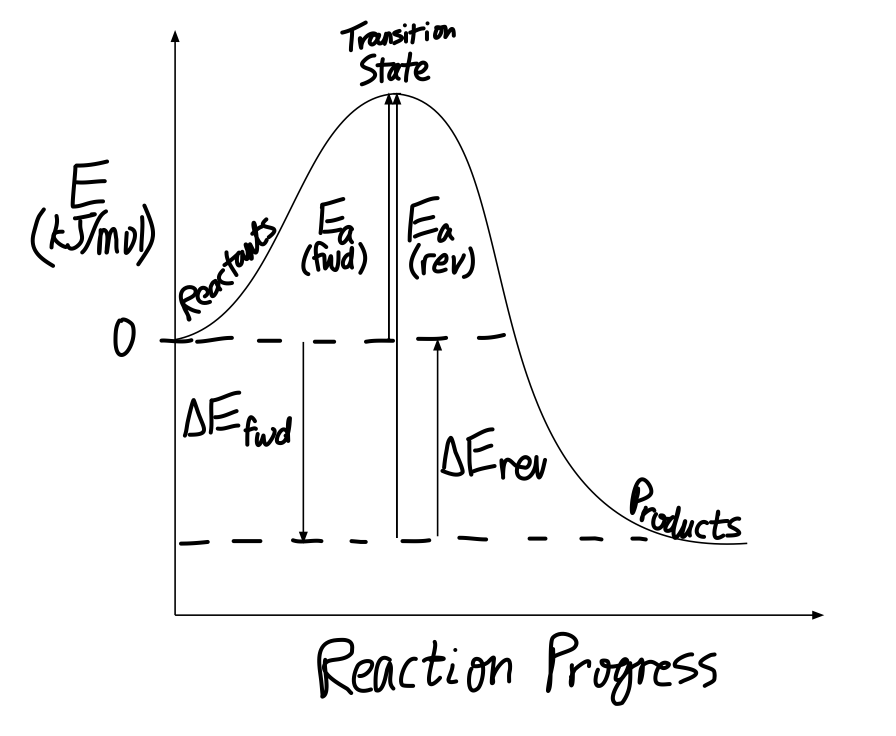
As they are, the values don't seem to make sense... there is an
Yet, if we have two reactions with activation energies
I'll let you work from the qualitative picture up above...

