A Bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells.
The first valence shell contains #2# electrons, and the second and third shell have #8# electrons each, and the number keeps growing.
To draw the Bohr diagram for #"NaCl"#, we should first draw the individual diagrams for both #"Na"# and #"Cl"#.
The atomic number of #"Na"# is #11#, so it has #11# electrons.
The first and second valence shells are completely full, since their #2# and #8# electrons only take up the first #10# of sodium's #11# electrons. Thus there will be #1# leftover electron in the third valence shell, so the Bohr diagram of #"Na"# can be drawn as follows:

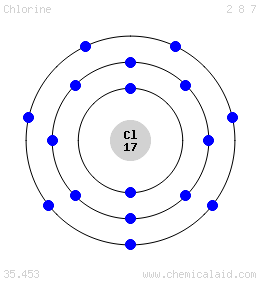
We can follow a similar process for chlorine, which has #17# electrons. The first two shells are full, taking up #10# of #"Cl"#'s #17# electrons, leaving #7# electrons in the third valence shell (remember that it has a capacity of #8#).
Now, we must draw the Bohr diagram for the #"NaCl"# model. #"NaCl"# is an ionic compound, meaning that electrons from each molecule that comprise the compound are ripped away from molecules and added to other molecules.
Notice that in #"Na"#, there is only #1# electron in the third valence shell. Because of this, #"Na"# wants to "shed" its single electron and form a #"Na"^+# ion. Similarly, #"Cl"# has #7# electrons of #8# possible in its third shell, so it will want to take an electron and form a #"Cl"^-# ion.
Hence the chlorine atom will take the electron in sodium's third valence shell and add it to its own, so the ionic compound would be drawn as:

Note:
#"Na"^+# has #10# electrons: its shells have #2# and #8# electrons, respectively.
#"Cl"^-# has #18# electrons: its shells have #2,8,# and #8# electrons, respectively.



