If #17/3 L# of a gas at room temperature exerts a pressure of #24 kPa# on its container, what pressure will the gas exert if the container's volume changes to #19/4 L#?
1 Answer
Aug 19, 2016
The gas will exert a pressure of
Explanation:
Let's begin by identifying our known and unknown variables.
The first volume we have is
We can obtain the answer using Boyle's Law:
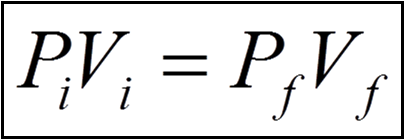
The letters i and f represent the initial and final conditions. All we have to do is rearrange the equation to solve for the final pressure.
We do this by dividing both sides by
Now all we do is plug in the values and we're done!

