In alpha decay, does a neutron decay into a proton and an electron.?
1 Answer
No, not quite.
Explanation:
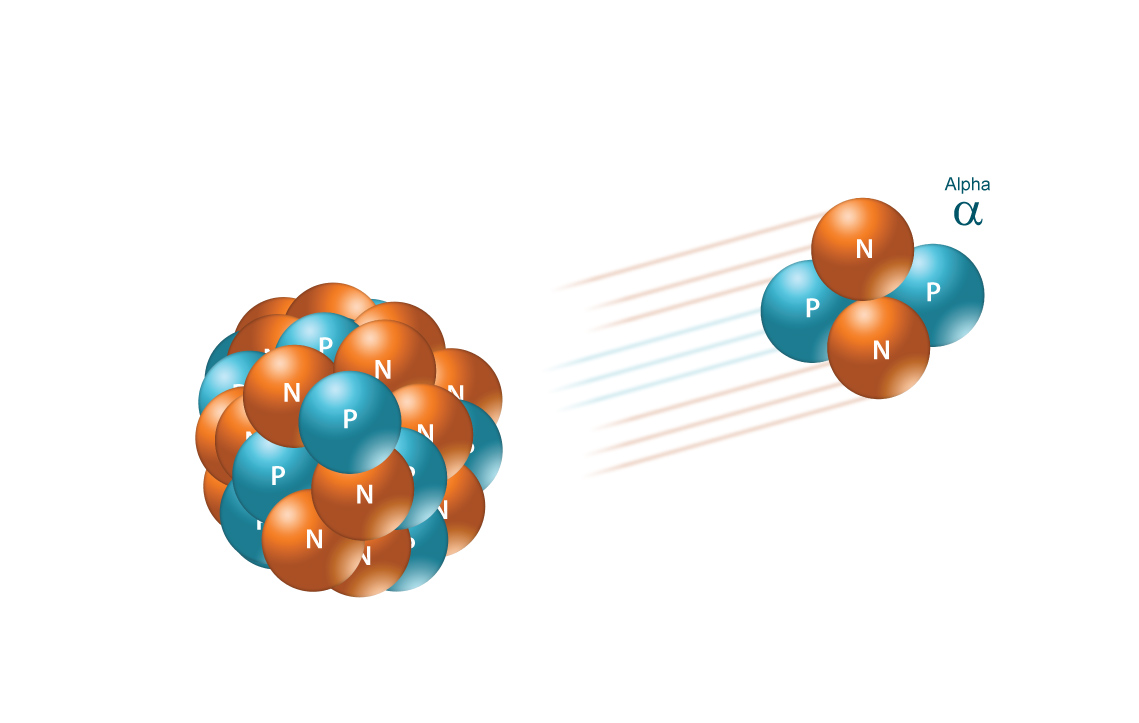
When a radioactive nuclide undergoes alpha decay, its nucleus of emits an alpha particle, which is essentially the nucleus of a helium-4 atom.
More specifically, an alpha particle contains
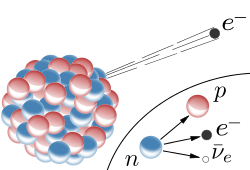
Now, what you're describing in the question is actually very close to being a beta decay, also known as a beta minus decay.
When a radioactive nuclide undergoes beta decay, one of its neutrons in being converted into a proton. At the same time, an electron, also called beta particle, and an electron antineutrino are emitted from the nucleus.
So remember
- alpha decay
#-># occurs when the nucleus emits an alpha particle
#color(red)(!)# Here nothing is being converted inside the nucleus
- beta decay
#-># occurs when the nucleus emits an electron and an electron antineutrino.
#color(red)(!)# Here a neutron is being converted into a proton and an electron