In the fourth dimension, is it possible to have a quadruple bond?
Ok so when you have two atoms sharing an orbital of electrons, they are singular bonded. If they share two orbitals, they're double bonded and if they share three, they're triple bonded. I've always learned in chemistry that there are at most 6 orbitals, each perpendicular to eachother that can be shared. Is it possible for, in the fourth dimension, another two orbitals be perpendicular to everything else? If so, could two four dimension atoms quadruple bond?
Is it possible in any way to have a quadruple bond?
Thanks!
Ok so when you have two atoms sharing an orbital of electrons, they are singular bonded. If they share two orbitals, they're double bonded and if they share three, they're triple bonded. I've always learned in chemistry that there are at most 6 orbitals, each perpendicular to eachother that can be shared. Is it possible for, in the fourth dimension, another two orbitals be perpendicular to everything else? If so, could two four dimension atoms quadruple bond?
Is it possible in any way to have a quadruple bond?
Thanks!
1 Answer
You do not even need four dimensions. Transition metals can sometimes form quadruple bonds in plain old three dimensional space, using
Explanation:
There is actually a Wikipedia article about this type of interaction (https://en.wikipedia.org/wiki/Quadruple_bond). An example is chromium (II) acetate, which would seem to be
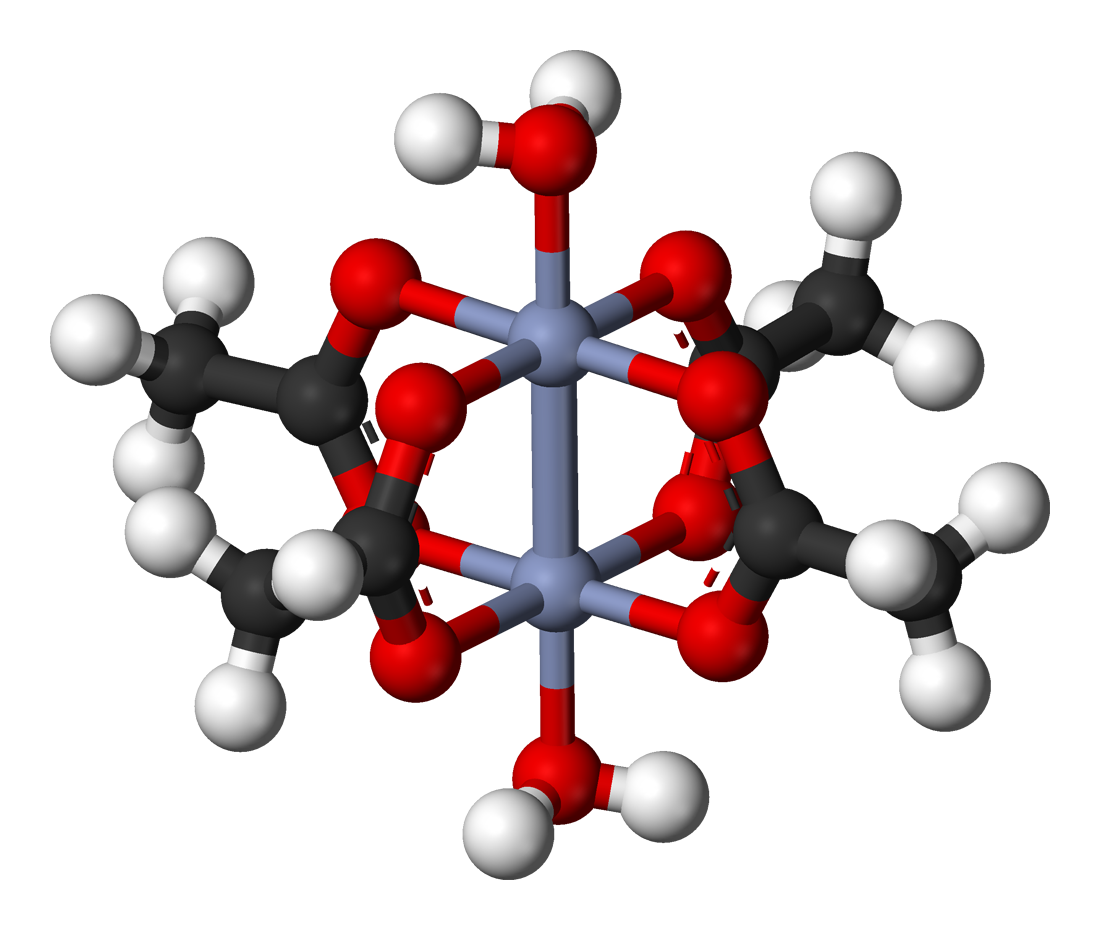 https://en.wikipedia.org/wiki/Quadruple_bond
https://en.wikipedia.org/wiki/Quadruple_bond
The quadruple bond between the chromium atoms in the center consists of these four bonds:
1) the usual sigma bond.
2) two pi bonds surrounding the sigma bond like the ones in molecular nitrogen or acetylene.
3) And, a delta bond. With the proper alignment of the surrounding oxygen atoms, a
Indeed, that extra delta bond is what holds the oxygen atoms in the proper alignment in the first place!


