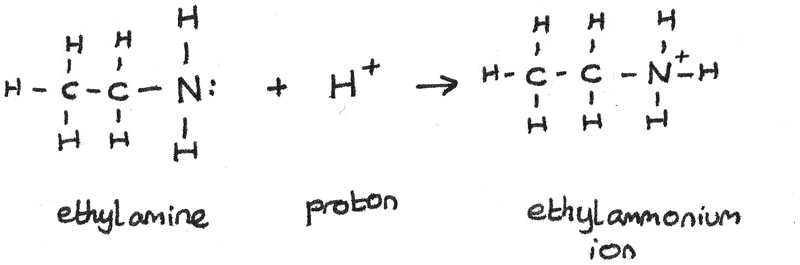
Is ethylamine a Bronsted-Lowry base?
1 Answer
May 5, 2015
Yes, ethylamine,
For example, when it reacts with water, ethylamine will accept a proton,
By definition, any compound that can act as a proton acceptor is a Bronsted-Lowry base.