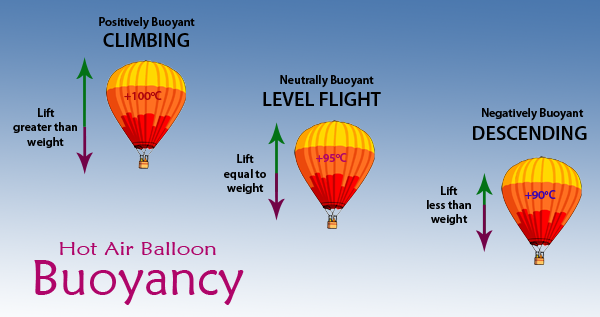
Is the density of the air in a heated hot air balloon less than the density of the air surrounding it and does that mean the volume of the same mass of air is increased within the balloon?
1 Answer
Yes and yes.
Explanation:
The density of the hot air trapped inside the balloon is indeed lower than the density of the air outside the balloon.
The reason for this is the direct relationship that exists between volume and temperature at constant pressure - this is known as Charles' Law.
Simply put, when you heat a gas, you essentially increase the average kinetic energy of its molecules. This implies that the molecules will now collide more often and with more energy, both between themselves and with the walls of the balloon.
Since pressure is held constant, meaning that you don't counteract these increasingly energetic collisions between the molecules and the walls of the balloon, the volume of the balloon will expand.
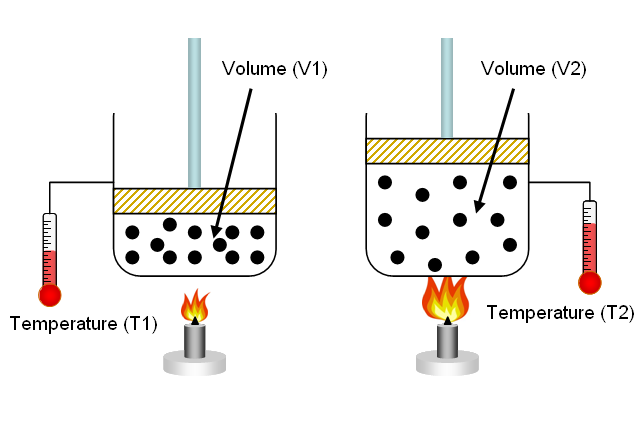
As a result, the molecules will now be further apart from each other inside the balloon, i.e. they wll be more spaced out.
Since density tells you the mass of the gas, which is presumed constant, per unit of volume, an increasing volume will result in a decreasing density.
From this point on, it's simply a matter of buoyancy.