The bond between which two atoms has the greatest degree of polarity in the following molecule?
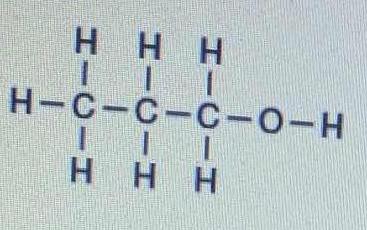
1 Answer
Jan 4, 2017
Between the oxygen and the hydrogen......
Explanation:
And how do we assess this? Probably the best metric is by
Pauling electronegativity of oxygen is

