The systematic (IUPAC) name for the amino acid threonine is (2S,3R)-2-amino-3-hydroxybutanoic acid. The systematic name indicates that threonine has two stereocenters, at positions 2 and 3, with S and R configurations, respectively. What is ths the Fischer projection of threonine. How many other possible stereoisomers of threonine are there?
1 Answer
Here's how I would draw the Fischer projection of threonine.
Step 1. Draw the structural formula of threonine.

Step 2. Draw the longest continuous chain of carbon atoms vertically, with
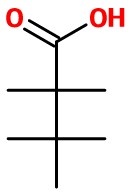
Step 3. Attach atoms to the bonds.
Arbitrarily put
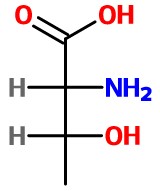
I have a 25 % chance of being right. If I am wrong, I will simply reverse the locations of the
Step 4. Determine the configuration at
The order of priority of the functional groups is
The sequence
But the
We want (S), so we must must interchange
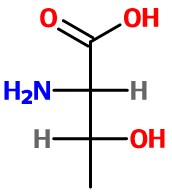
Step 4. Determine the configuration of
The order of priority of the functional groups is
The sequence
But the
The Fischer projection of threonine is therefore
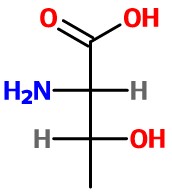
The number of possible stereoisomers is
Since threonine has 2 stereocentres, there are
So there are three other stereoisomers of threonine.

