The valence electron in potassium requires #6.95 xx 10^-19# #"J"# of energy to be removed. What frequency of light can do this? What type of electromagnetic radiation could do this?
1 Answer
Jul 3, 2017
Very blue visible photons or UV photons.
Explanation:
This electron in the
Now, referring to the EMR diagram that I gaurantee you most people don't have absolutely memorized:
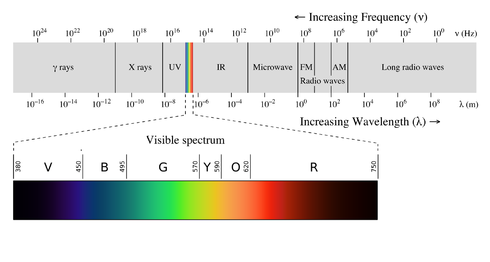
This energy corresponds with the frequency of very blue photons (visible) or UV photons.

