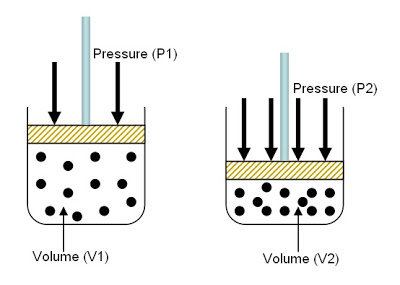
The volume of a gas is reduced from 4 L to 0.5 L while the temperature is held constant. How does the gas pressure change?
1 Answer
Sep 16, 2016
As the volume decreases, the pressure will increase.
Explanation:
In Boyle's law, which states that the volume and the pressure of a gas are inversely proportional as long as mass and temperature are kept constant.
So if the volume of the gas was decreased, the pressure would increase.