What are some examples of the properties of bases?
1 Answer
- They might want protons (Bronsted-Lowry definition)
- They might want to donate electrons (Lewis definition)
- They might donate
#"OH"^(-)# to solution (Arrhenius definition) - The conjugate base of a weak acid is a strong base
- The conjugate base of a strong acid is a weak base
A nice example of something that has most of the following properties is
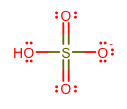
This base wants a proton according to the Bronsted-Lowry definition, and it will get that proton by donating electrons according to the Lewis definition, using the lone pairs on the
It is the conjugate base of
(Since the
#"pKa"# of#"H"_2"SO"_4# is about#1000# , it is reasonable to say that most#"H"_2"SO"_4# is actually deprotonated.)
Furthermore, although it might be difficult, it can be deprotonated to get
(This makes the conjugate base,
#"SO"_4^(2-)# , a strong base.)
It is not, however, a base by the Arrhenius definition, but an acid. It donates protons (

