What are some uses of UV/VIS spectroscopy?
1 Answer
UV/VIS spectroscopy is used for the quantitative determination of different substances.
The intensity of absorption at a given wavelength is related to the concentration by the Beer-Lambert Law:
Common analytes are transition metal ions, conjugated organic compounds, and biological molecules.
Transition metal ions
Solutions of transition metal ions can absorb visible light because electrons in the metal atoms can be excited from one electronic state to another.
http://upload.wikimedia.org/wikipedia/commons/thumb/7/7f/Cr(hexammine)3%2Bpng/300px-Cr(hexammine)3%2Bpng
The UV-visible spectrum of Cr(NH₃)₆³⁺ shows two weak transitions in the visible, corresponding to transitions between the d orbitals.
Conjugated Organic Compounds
Conjugated organic compounds absorb light UV/VIS light when their π electrons are excited to higher energy orbitals.
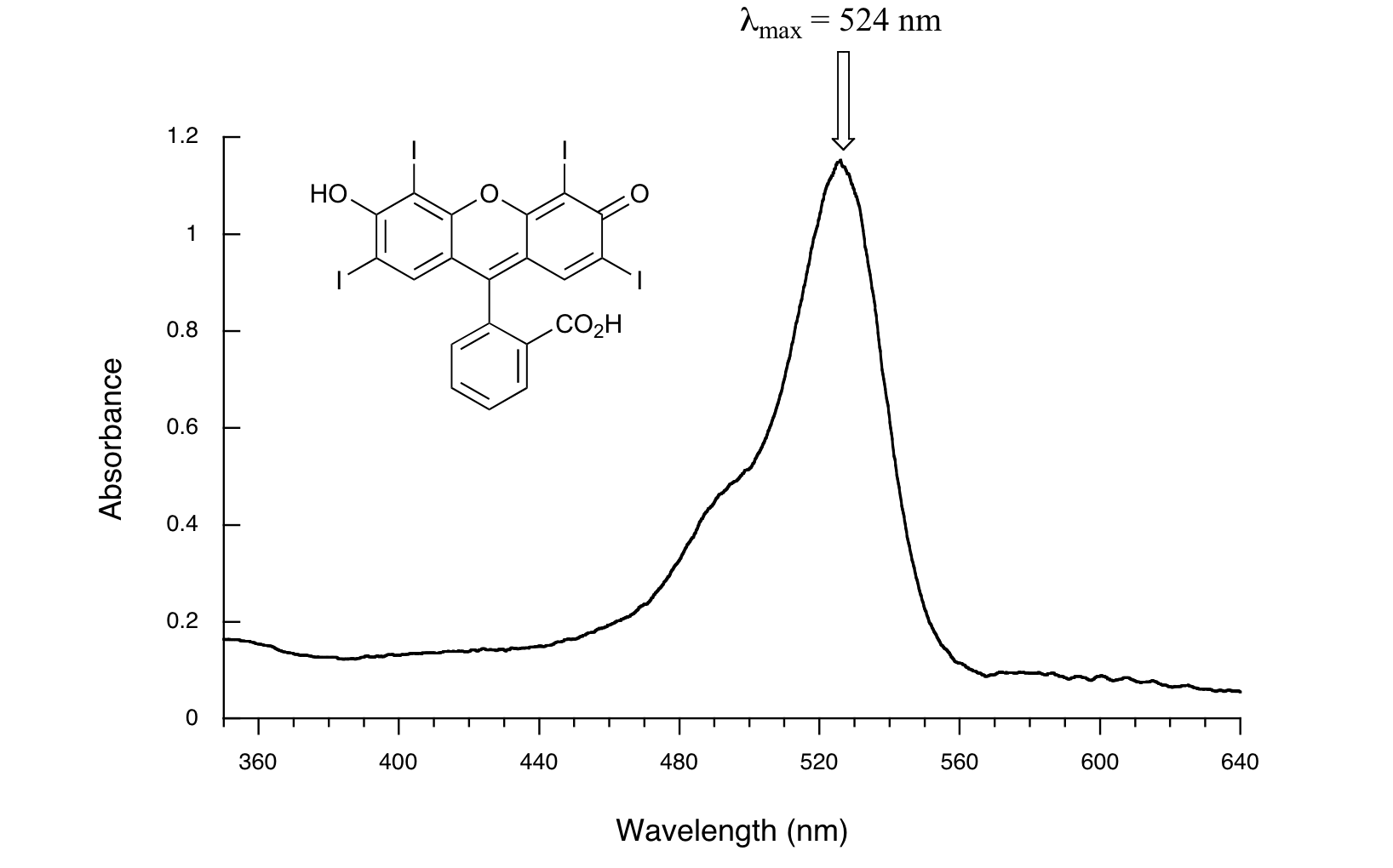
The highly conjugated food dye Red #3 absorbs at 524 nm.
 chemwiki.ucdavis.edu
chemwiki.ucdavis.edu
This is in the green region of the spectrum, so the compound appears red to our eyes.
Biological Molecules
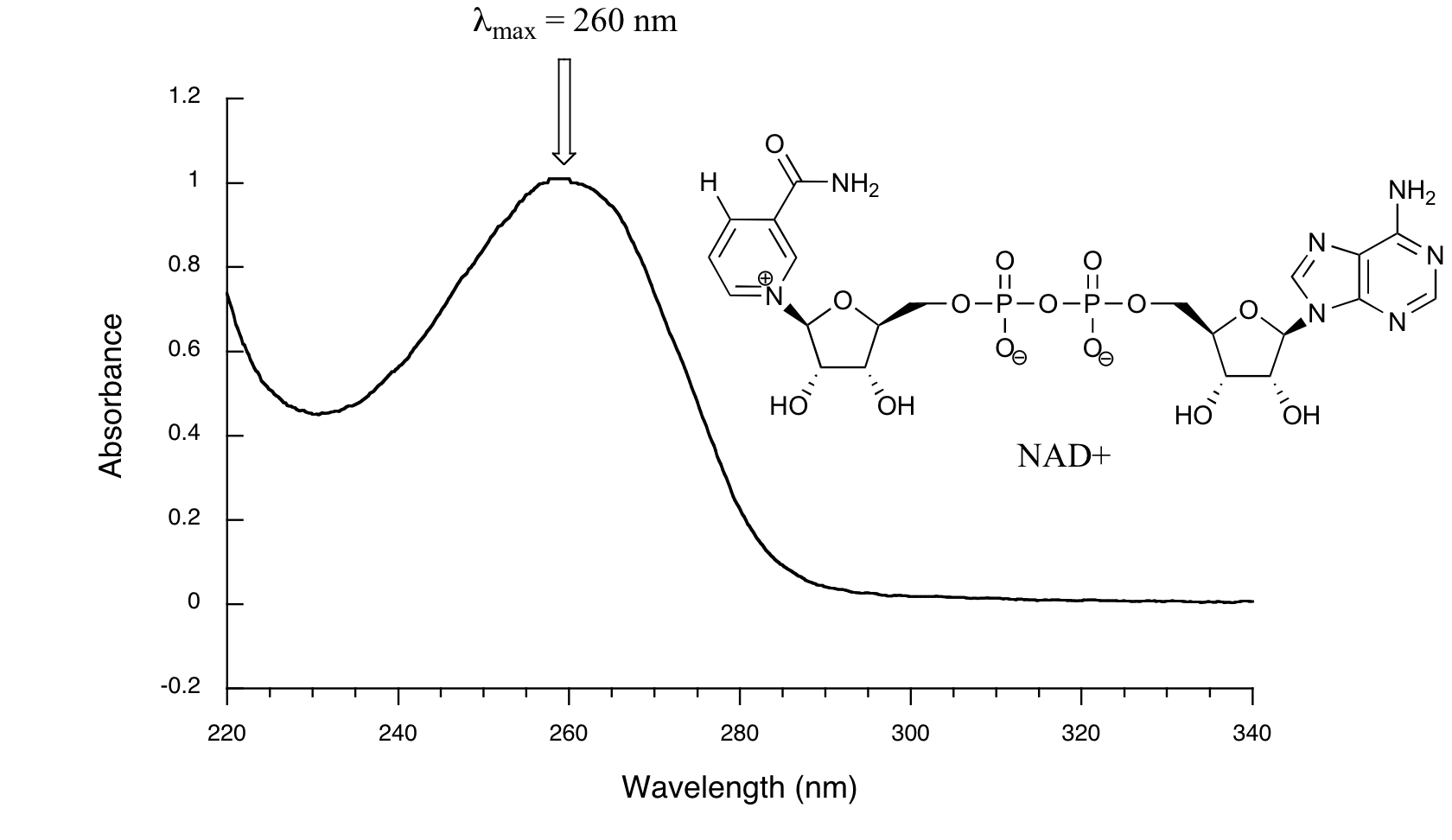
Nicotinamide adenine dinucleotide (NAD⁺) absorbs UV light at 260 nm due to its conjugated π systems.
 chemwiki.ucdavis.edu
chemwiki.ucdavis.edu
PRACTICAL APPLICATIONS
Some practical applications of UV/VIS spectrometry are:
- Quantitative analysis
- Monitoring chromatographic eluents
- Measuring reaction rates
- Measuring equilibrium constants
- Measuring film thickness
- Measuring spectra of astronomical features
- Measuring spectra of microscopic specimens

