What conditions make #DeltaG# always positive?
1 Answer
Sep 9, 2017
If ΔH is positive and ΔS is negative
Explanation:
If reaction is endothermic (ΔH is +ve), and entropy decreases (ΔS is -ve), then ΔG must be +ve and reaction is reactant-favored in the standard state.
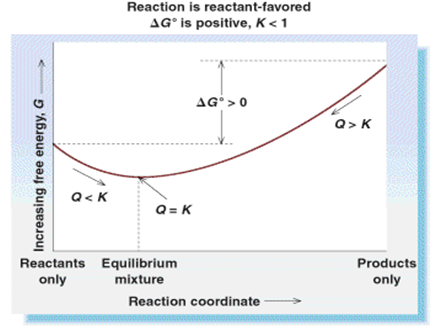
Where Q is the reaction quotient at that moment in time.

