What family of elements is relatively unreactive and why?
1 Answer
The noble gases because their valence shell is has 8 electrons.
Explanation:
Many chemical reactions take place because atoms don't have a full outer/valence electron shell. To achieve such, they lose, gain or share electrons in a chemical reaction.
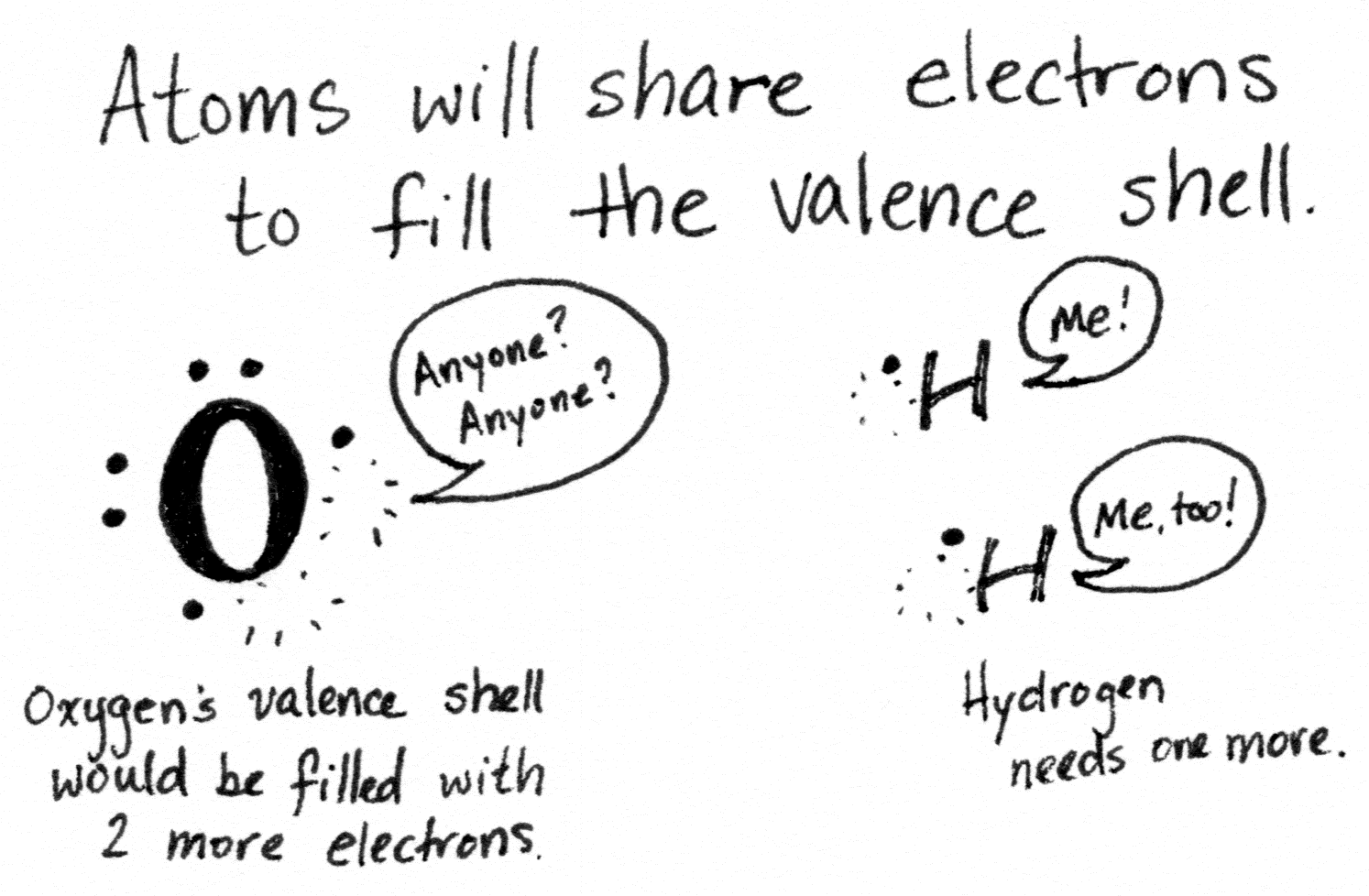
The noble gases all have full valence shells, and so they don't need to undergo a reaction to gain it. Hence, they are unreactive.



