What hybridization is involved in the carbon-carbon bonds?
1 Answer
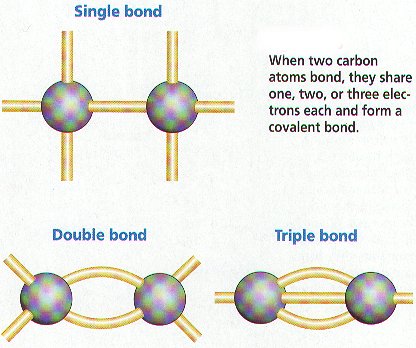
A carbon atom can form a covalent bond with another carbon atom, the most common way this is done being through a single bond (or a sigma bond).
In such a case, the sigma bond is formed between one hybridized orbital from each of the two carbon atoms.
In alkanes like ethane, carbon's orbitals are
In alkenes, carbon atoms can form double bonds; these bonds, consisting of one sigma and one pi bond, are formed with an
In alkynes, the carbon atoms can form triple bonds; these bonds, consisting of one sigma and two pi bonds, are formed with an