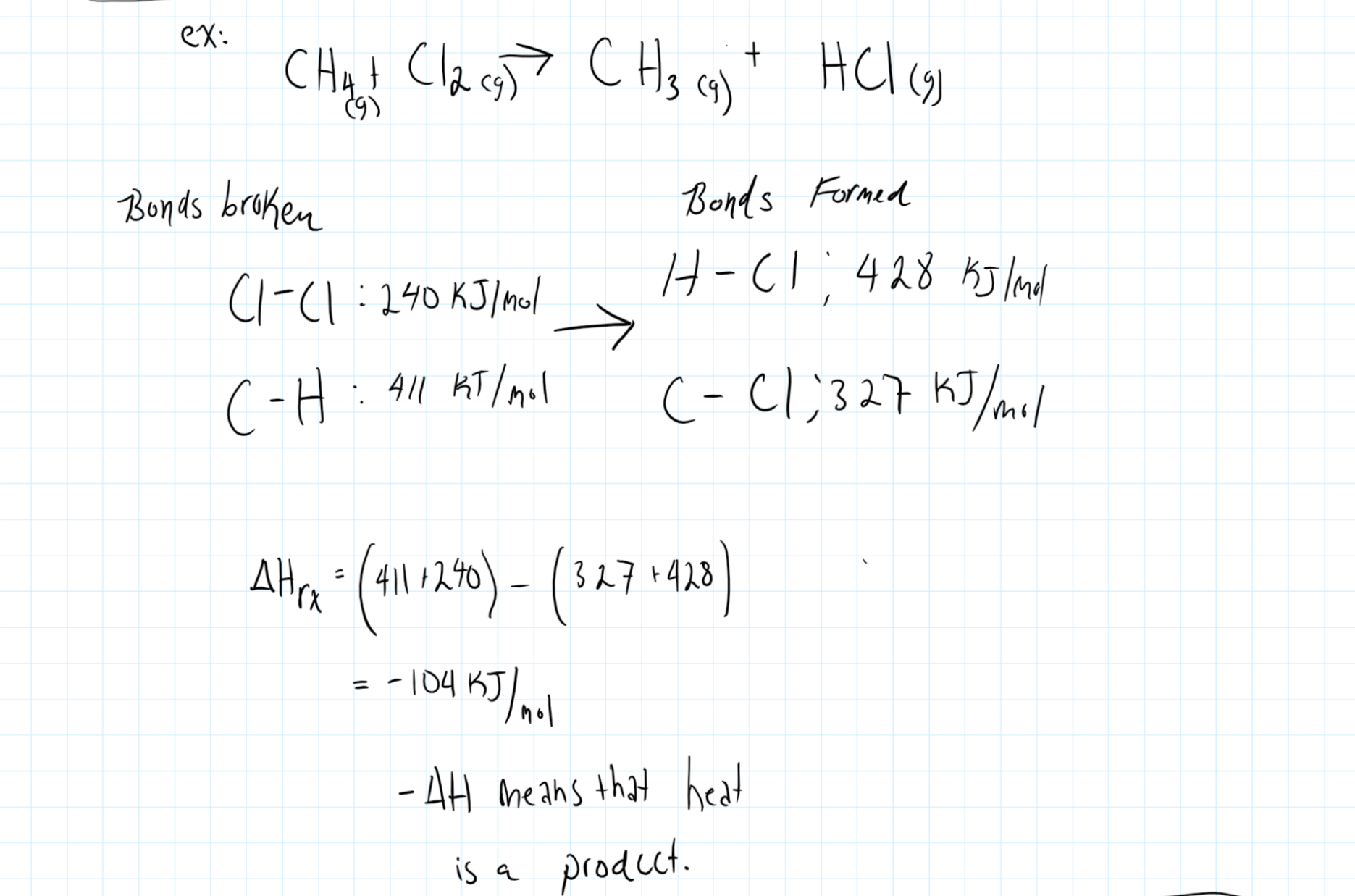What is a chemical reaction that absorbs heat from the surrounding? Does this reaction have a neutral, positive or negative #DeltaH# at constant pressure?
1 Answer
Dec 5, 2016
Negative
Explanation:
When energy is input into the system (heat)
Positive values of
When
Consider the graphic below where