What is a Lewis dot diagram?
1 Answer
Well, it is a diagram that represents the distribution of VALENCE electrons around atoms in a molecule...
Explanation:
The modern chemical bond is conceived to be region of high electron density between two positively charged atomic nuclei such that internuclear repulsion is negated and an attractive force results between the positively charged nuclei and the intervening electron cloud. The electrons surround individual atoms (and atoms bound in molecules) in orbitals whose shapes correspond to the Platonic solids, in such a way that the electron pairs are mutually repel each other....
And so it is important to account for the valence electronic configuration of an atom, as an atom, or as an atom in a molecule. And thus Lewis dot treatment distributes the valence electrons. And we can easily find the number of valence electrons for a given atom by noting its Group number in the Periodic Table, which number gives required the number of electrons.
For a simple example, consider ammonia,
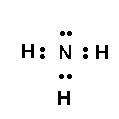
Nothing I say here will replace reading the relevant chapters in your text, so get to it...and we will be available for any questions that arise.

