What is a tetrahedral?
1 Answer
A tetrahedral is a shape in molecular geometry. It looks like this:
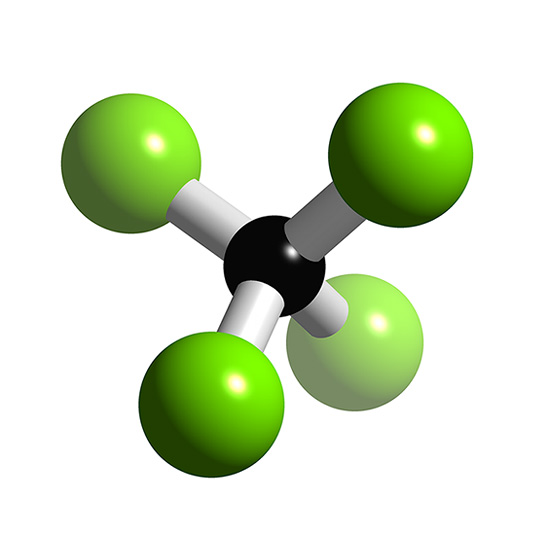
Tetrahedral shapes are formed by, according to electron domain geometry and VSEPR theory, four "electron domains" (bonds or lone electron pairs around the central atom). The tetrahedral shape is formed by the electrons repulsing one another, which forms the tetrahedron—the shape in which all the electrons are as far away from one another as possible.
Examples include methane (
Methane has a central carbon atom ringed by four hydrogens, which form the "spokes" of the tetrahedral molecule.
Tetrahedral molecules are nonpolar if the four surrounding atoms are the same, in the cases of methane and ammonium, and have a bond angle of 109.5˚.
However, tetrahedral molecules can also be polar if they are like chloroform (


